Accelerating Myofibrillar Repair: The Mechanistic Synergy of BPC-157 and MGF in Muscle Rupture Models
Explore the powerful synergy between BPC-157 and MGF in healing muscle ruptures. This article details the mechanisms of angiogenesis and satellite cell activation that accelerate recovery and reduce fibrosis.
Introduction: The Physiological Challenge of Muscle Rupture
In the field of regenerative biology, skeletal muscle rupture represents a complex physiological hurdle. Unlike minor micro-tears induced by standard hypertrophy training, frank muscle ruptures—whether partial or complete myotendinous junction failures—often result in sub-optimal recovery. The natural repair process, while robust, frequently prioritizes structural integrity over functional restoration. This results in fibrotic scar tissue replacement rather than the regeneration of contractile myofibrils, leading to a permanent reduction in force generation and a heightened risk of re-injury.
For researchers investigating peptide therapeutics, the goal is to shift this biological paradigm from "patching" to "regenerating." Two specific peptides have emerged as frontrunners in this localized repair environment: BPC-157 (Body Protection Compound-157) and MGF (Mechano Growth Factor). While both have demonstrated significant efficacy in isolation, their mechanisms of action are complementary rather than redundant.
Recent investigations into muscle trauma models suggest that combining the angiogenic and cytoprotective properties of BPC-157 with the satellite cell-activating characteristics of MGF creates a synergistic environment. This environment potentially accelerates myofibrillar repair while mitigating the formation of non-contractile collagen deposits. This article explores the mechanistic underpinnings of this synergy and presents a comprehensive overview of how these two peptides interact within the extracellular matrix of damaged tissue.
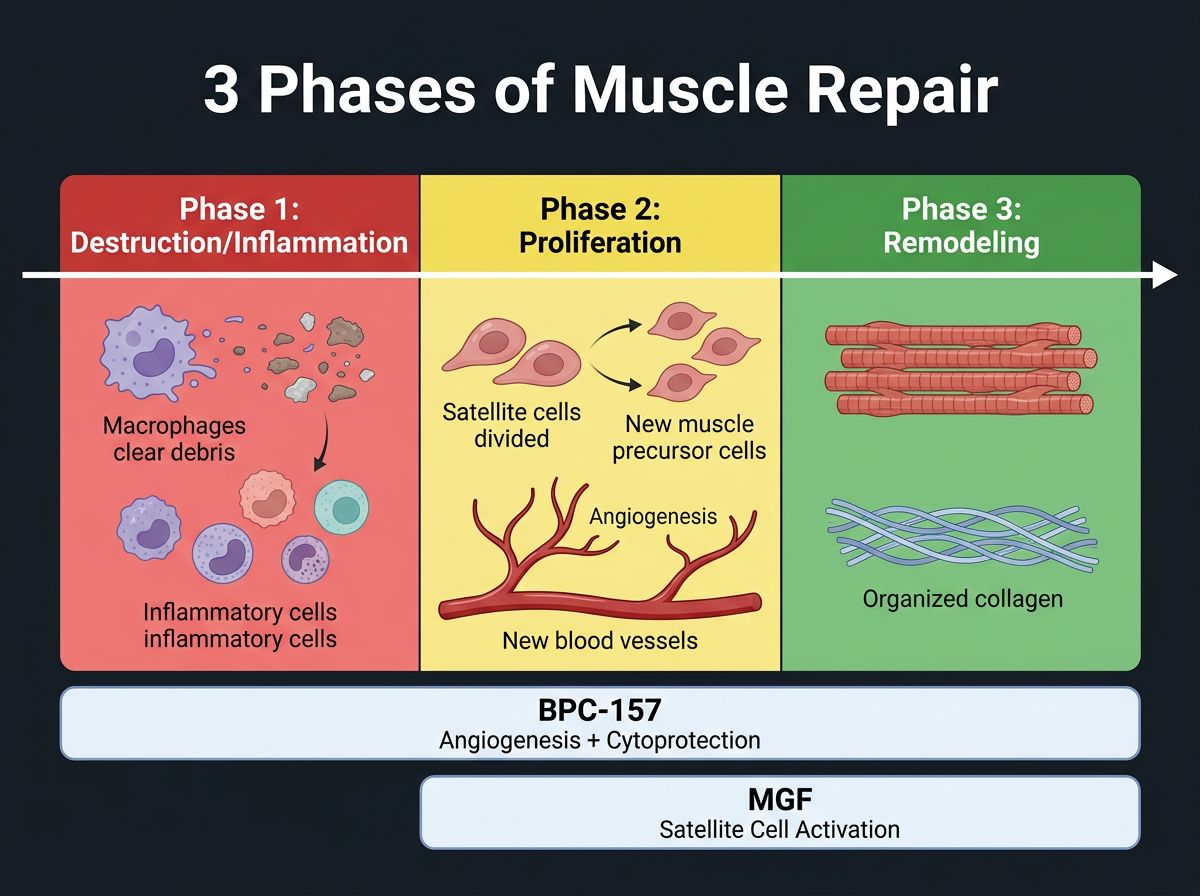
The Biology of Muscle Repair: Where Natural Healing Fails
To understand the utility of these research peptides, one must first delineate the standard phases of muscle healing. When a muscle fiber is severed, the body initiates a cascade of events categorized into three overlapping phases: destruction/inflammation, repair, and remodeling.
During the destruction phase, necrosis of the myofibers occurs, and the influx of calcium induces proteolysis. Inflammatory cells, particularly macrophages and neutrophils, invade the site to clear debris. While necessary, prolonged inflammation can be cytotoxic to the resident stem cells required for repair. The subsequent repair phase relies heavily on satellite cells—the muscle's distinct population of stem cells—to proliferate and fuse into myotubes.
However, the limiting factor in this process is often vascularization and the competition between muscle regeneration and fibrosis. Fibroblasts also migrate to the injury site, depositing collagen types I and III to restore tensile strength. Without adequate signaling to continue muscle differentiation, fibrosis wins out, creating a stiff, scar-filled muscle belly. This is the precise point of intervention for BPC-157 and MGF.
BPC-157: The Angiogenic Architect
BPC-157 is a stable pentadecapeptide derived from a protective protein found in human gastric juice. While its origins are gastric, its applications in research have proven systemic, with a profound affinity for soft tissue types including tendon, ligament, and skeletal muscle.
Mechanism of Action: The NO-VEGF Axis
The primary contribution of BPC-157 to muscle repair is its ability to modulate the production of nitric oxide (NO) and the expression of Vascular Endothelial Growth Factor (VEGF). In the context of a rupture, blood supply is invariably compromised. Ischemia (lack of oxygen) leads to cell death and prevents the transport of metabolic building blocks required for synthesis.
Research indicates that BPC-157 stimulates the expression of the KLF4 (Krüppel-like factor 4) gene, a transcription factor involved in endothelial cell growth. By upregulating VEGF production specifically at the site of injury, BPC-157 accelerates the formation of new capillary loops (angiogenesis). This re-vascularization is the non-negotiable prerequisite for healing; without a blood supply, satellite cells cannot proliferate.
Cytoprotection and Collagen Alignment
Beyond angiogenesis, BPC-157 exhibits direct cytoprotective effects. It has been shown to counteract the effects of various toxins and inflammatory agents. In muscle rupture models, BPC-157 does not merely speed up the deposition of collagen; it influences the organization of that collagen. Instead of the chaotic, cross-linked "haystack" pattern typical of scar tissue, BPC-157 promotes a more parallel alignment of collagen fibers, more closely mimicking the original tendon urgency. This is critical for retaining the load-bearing capacity of the muscle.
Furthermore, BPC-157 is frequently studied alongside TB-500 (Thymosin Beta-4), another peptide involved in actin sequestration. While TB-500 aids in cell migration, BPC-157 provides the angiogenic foundation that makes migration possible.
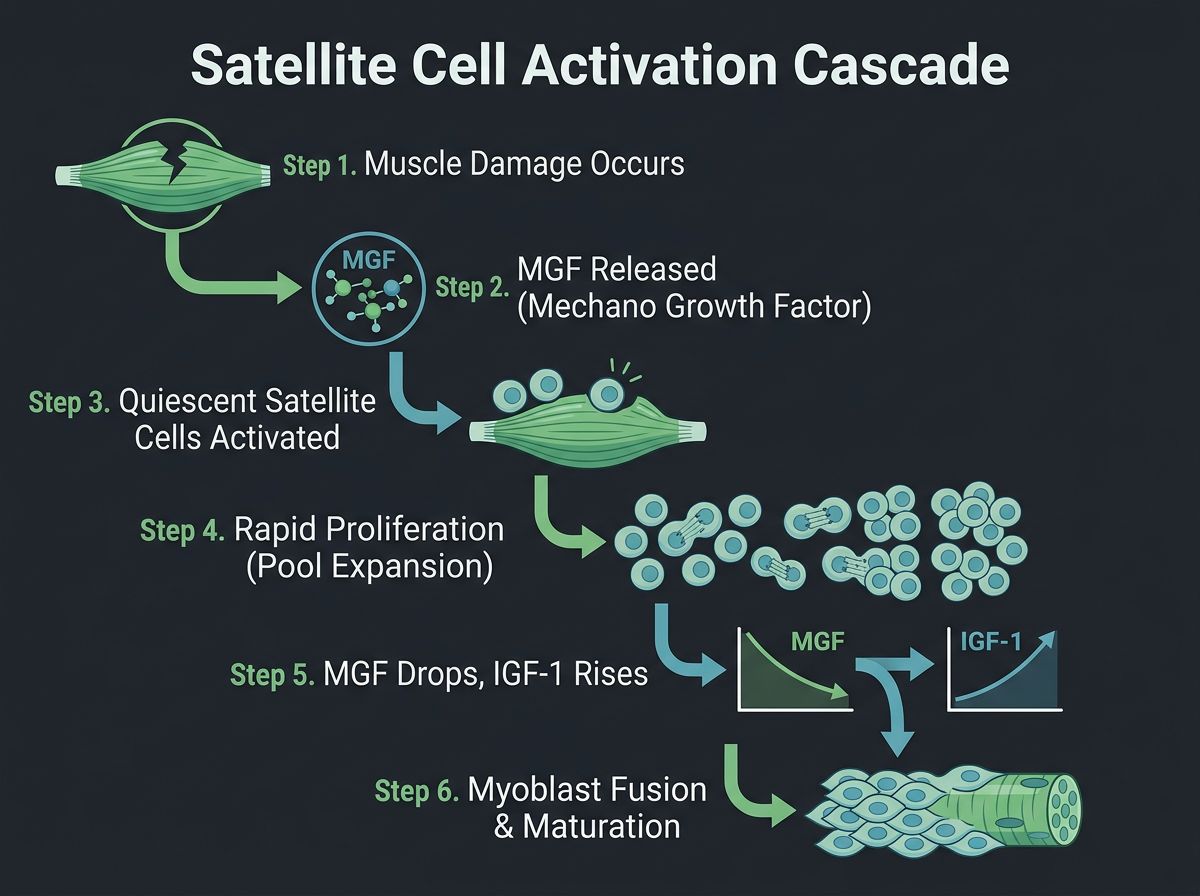
MGF: The Satellite Cell Activator
While BPC-157 builds the "roadway" (blood vessels) and protects the tissue, MGF provides the "construction crew" (muscle cells). MGF is a splice variant of the Insulin-Like Growth Factor-1 (IGF-1) gene. It is naturally expressed in muscle tissue immediately following mechanical overload or damage, hence the name Mechano Growth Factor.
MGF vs. IGF-1Ea: A Crucial Distinction
It is vital to distinguish between the common systemic IGF-1 (often called IGF-1Ea) and MGF (also known as IGF-1Ec in humans). Systemic IGF-1 is largely produced by the liver and is involved in protein synthesis and the maturation of muscle fibers. MGF, conversely, acts earlier in the chain.
MGF is the acute responder. Its primary biological role is to activate quiescent satellite cells. Upon activation, these stem cells re-enter the cell cycle and proliferate exponentially to create a pool of myoblasts. Crucially, MGF prevents these cells from differentiating (fusing) too early. It essentially expands the "pool" of available repair material. Once the pool is sufficiently large, MGF levels drop, and IGF-1Ea levels rise to undergo fusion and maturation.
The Stability Issue: Enter PEG-MGF
Synthetic MGF has a very short half-life in the body—often measured in minutes—due to rapid breakdown by enzymes. For research purposes demanding sustained bioavailability, scientists utilize PEG MGF (Pegylated Mechano Growth Factor). The process of pegylation involves attaching a polyethylene glycol polymer to the peptide. This modification surrounds the peptide in a protective shell, shielding it from enzymatic degradation and extending its half-life from minutes to days. This allows for a single administration to provide sustained satellite cell activation.
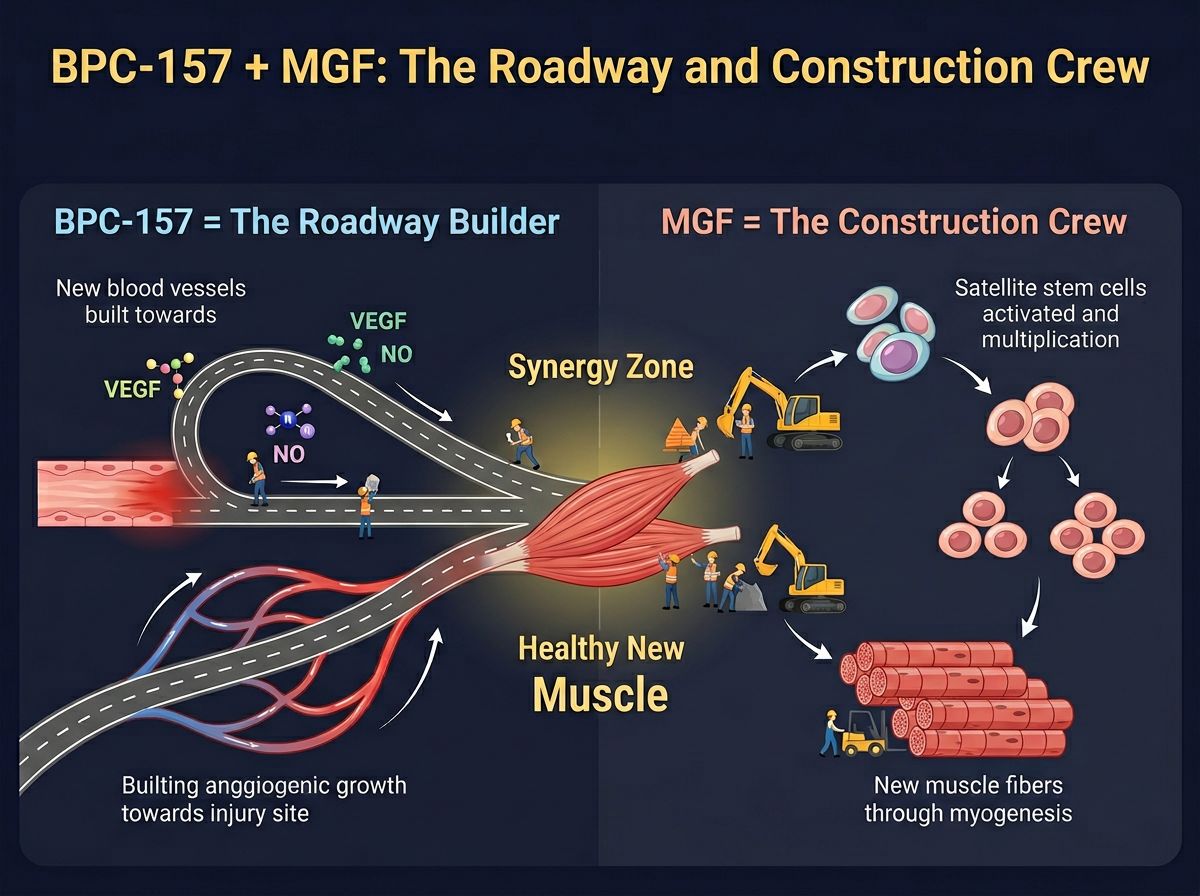
The Mechanistic Synergy: Why Use Both?
The combination of BPC-157 and MGF addresses the two primary bottlenecks of muscle healing: ischemia (addressed by BPC-157) and stem cell depletion (addressed by MGF). When administered concurrently in animal models, these peptides engage in a complementary feedback loop that accelerates functional recovery beyond what either could achieve alone.
1. Metabolic Support for Hyper-Proliferation
MGF is a potent mitogen; it signals cells to divide rapidly. This cellular proliferation creates a massive metabolic demand. Dividing cells require glucose, amino acids, and oxygen. In a ruptured muscle with compromised vasculature, MGF alone might trigger proliferation that the local environment cannot support, leading to apoptosis (cell death) of the newly formed myoblasts.
BPC-157 resolves this by rapidly re-establishing the capillary network. By increasing the density of blood vessels via the VEGF pathway, BPC-157 ensures that the satellite cell pool activated by MGF receives the nutrients required to survive and thrive. This is the physiological definition of synergy: BPC-157 fuels the engine that MGF starts.
2. Modulation of the Fibrotic Response
A major challenge in muscle rupture is the race between myogenesis (regaining muscle) and fibrogenesis (forming scar tissue). Through the upregulation of specific growth factors, BPC-157 tends to dampen the excessive inflammatory response that drives fibrotic scarring. Meanwhile, MGF recruits mesenchymal stem cells but directs them toward a myogenic lineage rather than a fibrotic one.
Research suggests that the co-presence of these peptides results in a healed defect that contains a higher ratio of contractile muscle fibers to collagen deposits. This translates to a muscle that is not only "healed" but also "functional," maintaining its ability to stretch and contract without re-tearing at the scar site.
| Healing Phase | Role of BPC-157 | Role of MGF (PEG-MGF) | Synergistic Outcome |
|---|---|---|---|
| Inflammation/Destruction | Modulates inflammatory cytokines; provides cytoprotection to surviving tissue. | Signals acute repair response; recruiting immune cells to clear debris. | Reduced tissue necrosis and faster transition to repair phase. |
| Proliferation | Stimulates VEGF for angiogenesis (new blood vessels) to feed the site. | Activates quiescent satellite cells to divide and multiply continuously. | Massive expansion of myoblast pool supported by adequate blood supply. |
| Remodeling | Aligns collagen fibers parallel to force lines; strengthens connective tissue. | Allows myoblast fusion; prevents premature differentiation until density is high. | Restored contractile strength with minimal fibrotic scar tissue/stiffness. |
Research Applications and Protocol Considerations
In laboratory settings, the administration of these peptides is typically studied via subcutaneous or intramuscular injection near the site of injury. It is crucial to note that while systemic administration (subcutaneous injection away from the injury) is effective for BPC-157 due to its stability, MGF is often studied for its local effects.
Often, researchers will look toward blends or "stacks" involving other remodeling agents. For example, GHK-Cu is a copper peptide noted for its ability to remodel collagen and skin, often used in conjunction with BPC-157 during the late stages of healing to reduce surface scarring. Similarly, BPC-157 + TB-500 Blend is a common starting point in research, with MGF added specifically when muscle tissue (myofibril) damage is the primary concern.
Timing the Administration
Based on the biological half-lives and mechanisms:
- BPC-157: Generally administered daily or twice daily in research protocols to maintain high levels of the peptide during the acute angiogenic phase.
- PEG MGF: Due to the extended half-life provided by pegylation, research protocols often call for less frequent administration (e.g., every 48 to 72 hours). Over-saturation of MGF receptors can potentially lead to downregulation, so pulsed administration mimics the natural pulsatile release of growth factors.
The Role of Quality Control in Peptide Research
The efficacy of these peptides in a research setting is entirely dependent on the purity of the synthesis. Contaminants such as trifluoroacetic acid (TFA) or incomplete peptide sequences can induce inflammation, counteracting the very healing properties being studied. Ensuring the peptide source utilizes rigorous high-performance liquid chromatography (HPLC) and mass spectrometry is vital.
At Alpha Carbon Labs, we emphasize transparency in our peptide synthesis processes. Researchers should always verify the integrity of their compounds through valid COA Documents before commencing any live-subject or cellular study.
Expanded Mechanistic Review: The Signaling Pathways
To fully appreciate the synergy, one must look at the intracellular signaling pathways impacted by these peptides. BPC-157 has been observed to interact with the FAK-paxillin pathway. Focal Adhesion Kinase (FAK) is critical for cell migration and adhesion. By stimulating FAK phosphorylation, BPC-157 essentially tells cells where to go and how to stick together to close a wound.
Simultaneously, MGF activates the MAPK/ERK pathway. This is the canonical pathway for cell division. Interestingly, MGF has a unique C-terminal E-domain that is not present in systemic IGF-1. This E-domain is believed to be responsible for the specific ability of MGF to keep cells in a proliferative state. Using IGF-1LR3 (Long Arg3 IGF-1) too early in a muscle rupture model can invoke premature differentiation, resulting in smaller, weaker muscles. MGF delays this differentiation, allowing for a larger tissue volume to be generated. The transition from MGF dominance to IGF-1 dominance is a delicate biological handover that dictates the final size of the repaired muscle.
Implications for Tendon and Ligament Interface
Muscle ruptures rarely occur in the "red" meat of the muscle belly alone; they frequently involve the myotendinous junction (MTJ). The MTJ is the interface where contractile muscle fibers mesh with the stiff collagen of the tendon. This zone is notoriously difficult to heal due to the difference in tissue compliance.
BPC-157 shines in this specific arena. Studies involving rat Achilles tendon transections have shown that BPC-157 significantly increases the load-to-failure strength of the healing tendon. When combined with MGF, which ensures the muscle side of the junction remains healthy and contractile, the integrity of the total biomechanical unit is preserved. This is relevant for research into complex injuries where muscle tears away from the bone or tendon.
For researchers focusing specifically on the connective tissue aspect rather than the muscle belly, exploring peptides like CJC-1295 or Ipamorelin may be relevant, as they increase systemic Growth Hormone, which increases collagen synthesis. However, for direct, localized repair of the rupture itself, the BPC-157 and MGF combination remains the gold standard in current literature.
Safety and Future Research Trends
Current literature on BPC-157 and MGF emphasizes their high safety profile in animal models. BPC-157, being a native gastric peptide derivative, shows virtually no toxicity even at massive dosages. MGF, while potent, acts locally (autocrine/paracrine signaling), minimizing the risk of systemic organ growth that is sometimes feared with high-dose systemic IGF-1 administration.
Future research is drifting toward advanced delivery systems. Hydrogels impregnated with BPC-157 and MGF are currently being investigated. These gels would be injected into a rupture site, polymerizing to form a scaffold that slowly releases the peptides over weeks. This would mimic the extracellular matrix and provide a physical structure for cells to climb across, further accelerating the bridging of large muscle gaps.
Conclusion
The synergy between BPC-157 and MGF represents a sophisticated approach to biological engineering. By pairing a potent angiogenic and cytoprotective agent (BPC-157) with a dedicated muscle stem cell activator (MGF), researchers can theoretically bypass the natural limitations of the mammalian healing response. This combination facilitates a repair process that is faster, less fibrotic, and functionally superior to natural healing.
As our understanding of peptide signaling pathways evolves, the dual-administration protocol of these agents is likely to become a cornerstone of soft tissue trauma research. For the scientific community, the focus remains on optimizing dosing schedules and delivery vectors to fully harness the regenerative potential of these remarkable peptides.
References
- 1. Sikirić, P., et al. (2010). 'BPC 157 and blood vessels.' Current Pharmaceutical Design, 16(10), 1224-1234.
- 2. Goldspink, G. (2005). 'Mechanical signals, IGF-I gene splicing, and muscle adaptation.' Physiology, 20(4), 232-238.
- 3. Chang, C. H., et al. (2011). 'The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration.' Journal of Applied Physiology, 110(3), 774-780.
- 4. Kandalla, P. K., et al. (2011). 'MGF: A peptide variant of IGF-1 that acts as a mechano-sensitive/local autocrine growth factor.' Frontiers in Endocrinology, 2.
- 5. Sikirić, P., et al. (2006). 'Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts.' Molecules, 11(11), 846-855.
- 6. Hwang, J. H., et al. (2009). 'Local expression of IGF-I splice variants in regenerating muscle of the rat.' Journal of Orthopaedic Research, 27(9), 1195-1200.
- 7. Pevec, D., et al. (2010). 'Impact of pentadecapeptide BPC 157 on muscle healing impaired by systemic corticosteroid application.' Medical Science Monitor, 16(3), BR81-BR88.
- 8. Mills, P., et al. (2007). 'Mechano growth factor, but not insulin-like growth factor-1, recruits mesenchymal stem cells to sites of tissue damage.' Journal of Biological Chemistry.
- 9. Vukojević, J., et al. (2018). 'Pentadecapeptide BPC 157 and the central nervous system.' Neural Regeneration Research, 13(5), 823.
- 10. Atalay, M., et al. (2016). 'Mechano Growth Factor (MGF) E peptide promotes proliferation and inhibits differentiation of myoblasts.' Growth Factors, 34.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.