The Anti-Inflammatory Axis: Investigating the Combined Potential of KPV and BPC-157 in Epithelial Research
Explore the synergistic effects of KPV and BPC-157 peptides in epithelial research, focusing on their mechanisms for resolving inflammation and accelerating tissue repair in gastrointestinal and dermatological models.
The Anti-Inflammatory Axis: Investigating the Combined Potential of KPV and BPC-157 in Epithelial Research
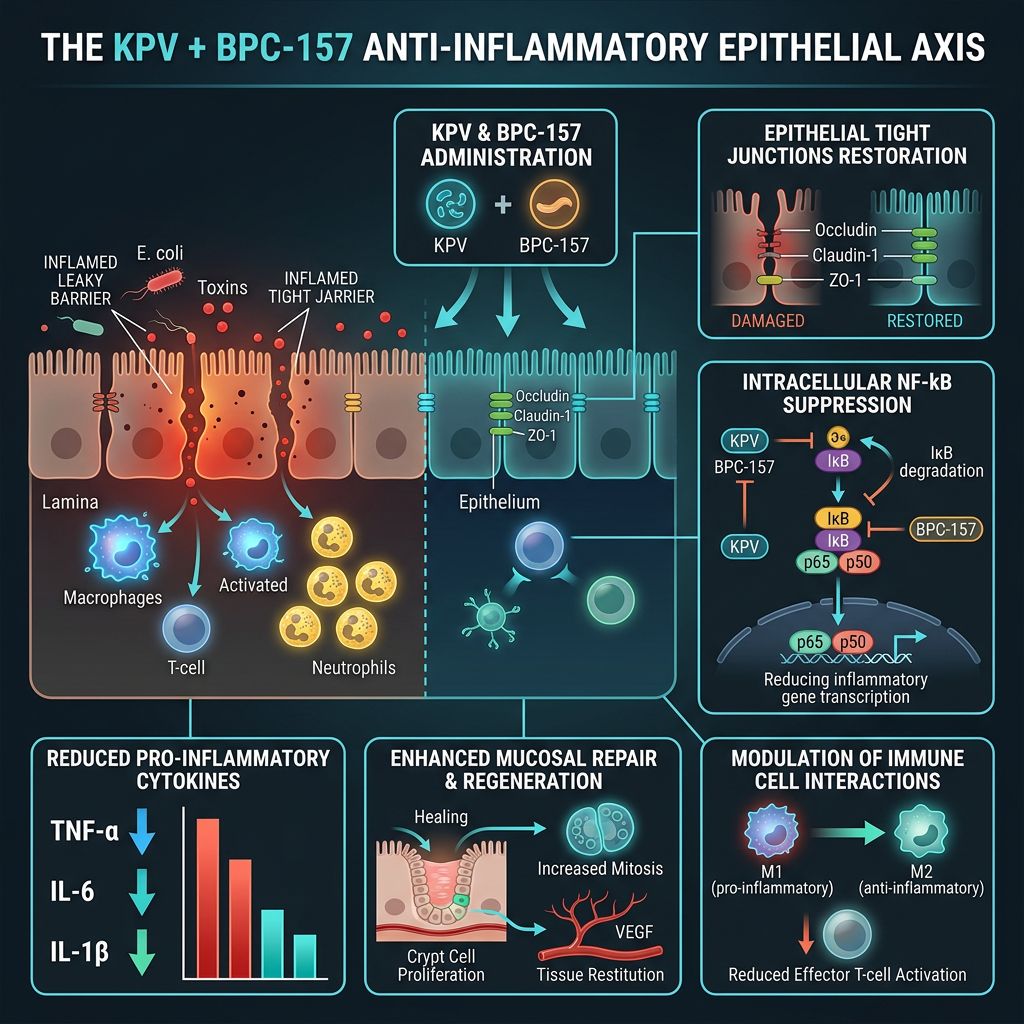
The field of regenerative medicine and inflammatory research has seen a paradigm shift with the emergence of targeted peptide therapies. Among these, the combination of KPV (Lysine-Proline-Valine) and BPC-157 (Body Protection Compound 157) represents a particularly potent research axis. While BPC-157 is widely recognized for its systemic cytoprotective actions and angiogenic modulation, KPV—a C-terminal tripeptide fragment of alpha-Melanocyte Stimulating Hormone (α-MSH)—offers a highly specialized anti-inflammatory mechanism that targets the NF-κB signaling pathway directly within epithelial cells.
Researchers are increasingly investigating how these two compounds, when utilized in tandem, might provide a synergistic approach to resolving chronic inflammation and accelerating barrier repair in the gut, skin, and mucosal tissues. This article explores the molecular intricacies of this "Anti-Inflammatory Axis," evaluating the existing data on their co-administration in regenerative models.
The Molecular Profile of KPV: The Tripeptide Powerhouse
KPV is a naturally occurring tripeptide that exerts its effects through the melanocortin receptors, though research suggests its anti-inflammatory properties may also be mediated through receptor-independent pathways. Unlike larger melanocortin peptides, KPV does not typically induce pigmentary changes, making it an ideal candidate for research focused purely on inflammatory modulation.
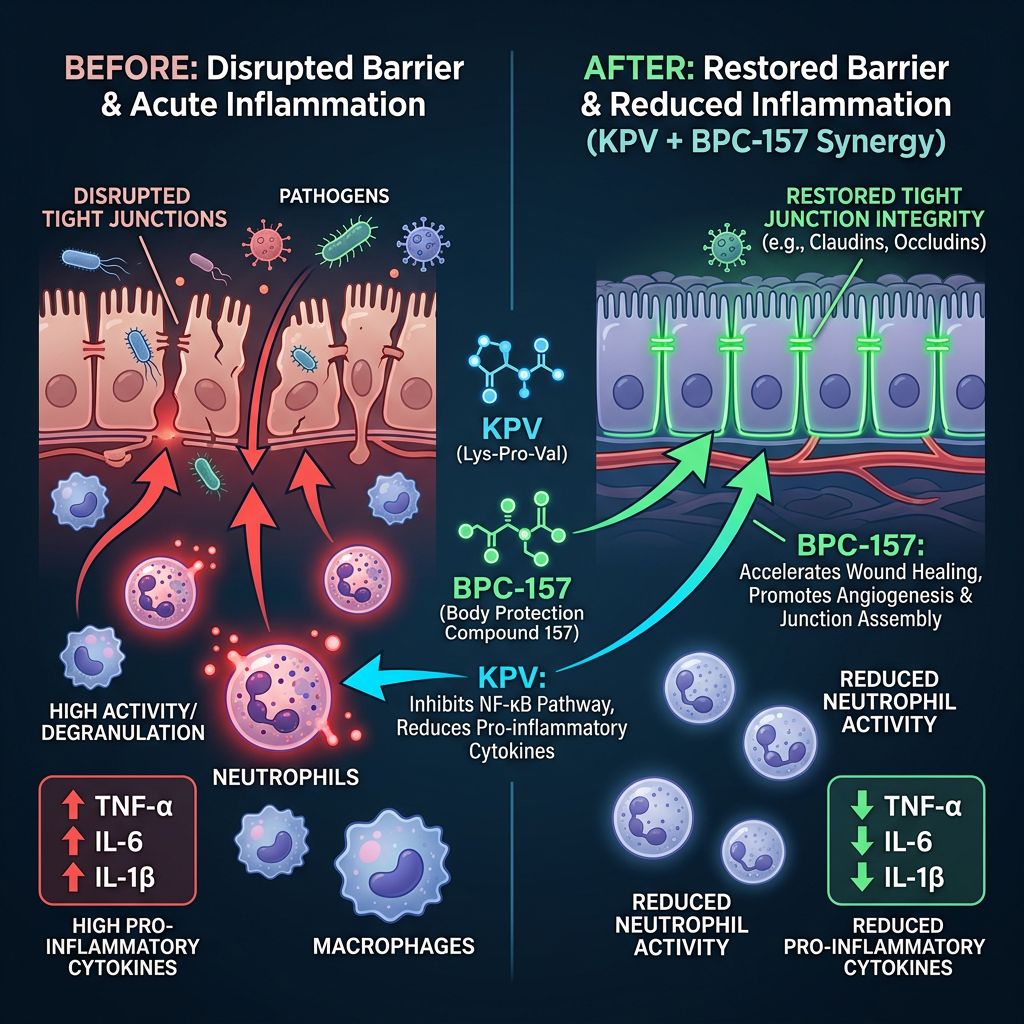
The primary mechanism of KPV involves the inhibition of the Nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-κB). NF-κB is a central mediator of the inflammatory response; its activation leads to the transcription of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. KPV enters cells through specialized transporters, such as PepT1, which is upregulated in inflamed intestinal tissues. Once inside, it inhibits the translocation of NF-κB to the nucleus, effectively cutting off the inflammatory signal at its source.
BPC-157: The Master of Cytoprotection
BPC-157 is a 15-amino acid sequence derived from human gastric juice. Its stability in unfavorable environments—such as the highly acidic gastric lumen—is a hallmark of its design. Over decades of research, BPC-157 has demonstrated an unparalleled ability to promote "healing from within" by modulating growth factors and promoting the formation of new blood vessels through VEGFR2 expression.
Unlike traditional anti-inflammatories that may inhibit healing as a side effect (such as corticosteroids), BPC-157 facilitates a pro-angiogenic environment that supports tissue reconstruction while simultaneously downregulating systemic inflammatory markers. It acts as a bridge, ensuring that as inflammation is lowered, the structural integrity of the tissue is actively restored.
Synergy in Gastrointestinal Research
The gastrointestinal (GI) tract is one of the most complex environments for inflammatory research due to the delicate balance between the microbiome, the mucosal barrier, and the immune system. The combination of KPV and BPC-157 offers a multi-target strategy for addressing Inflammatory Bowel Disease (IBD) and Leaky Gut Syndrome models.
- KPV in the Gut: Research published in Gastroenterology highlights that KPV acts directly on intestinal epithelial cells to reduce the production of pro-inflammatory cytokines. By utilizing the PepT1 transporter, KPV concentrates precisely where the inflammation is most acute.
- BPC-157 in the Gut: BPC-157 has been shown to counteract the damage caused by NSAIDs, alcohol, and stress-induced ulcers. It promotes the healing of the "gut-brain axis" and stabilizes the intestinal basement membrane.
When researchers utilize a BPC-157 + TB-500 Blend or combine KPV with BPC-157, they target two distinct phases of recovery. KPV halts the active recruitment of inflammatory cells and the cytokine storm, while BPC-157 initiates the repair of the tight junctions and cellular matrix. This dual action is critical in models where epithelial permeability is compromised.
Applications in Dermatological and Wound Healing Models
Beyond the GI tract, the skin is a primary site for KPV and BPC-157 research. Epithelial integrity in the skin is often compromised by chronic wounds, dermatitis, or burns. KPV’s ability to inhibit TNF-α is particularly relevant here, as TNF-α is a primary driver of chronic skin inflammation.
In dermatological studies, KPV has been observed to reduce edema and infiltrate of inflammatory cells into the dermis. When paired with BPC-157—which stimulates fibroblast migration and collagen synthesis—the rate of wound enclosure in animal models is significantly accelerated. This synergy is often compared to the effects seen in the BPC-157 + TB-500 + GHK-Cu Blend, which emphasizes both healing and structural remodeling.
Comparative Analysis: KPV vs. BPC-157
| Feature | KPV | BPC-157 |
|---|---|---|
| Primary Mechanism | NF-κB pathway inhibition & cytokine reduction | VEGFR2 modulation & growth factor upregulation |
| Standard Receptor Target | Melanocortin receptors (MC1R) or PepT1 transport | Modulates Nitric Oxide (NO) and Collagen pathways |
| Epithelial Impact | Reduces inflammatory signaling and oxidative stress | Promotes angiogenesis and basement membrane repair |
| Organ Specificity | High affinity for Intestine and Skin | Systemic, but highly effective in GI and Musculoskeletal |
| Research Focus | Autoimmune/Allergic Inflammation | Tissue Trauma and Degenerative Disease |
The Role of Quality Control in Peptide Research
For researchers investigating these pathways, the purity of the compounds is paramount. Small variations in peptide sequences or the presence of trifluoroacetic acid (TFA) salts can alter the cellular response, especially in the sensitive NF-κB pathways targeted by KPV. At Alpha Carbon Labs, we ensure that every batch of KPV and BPC-157 undergoes rigorous testing. Researchers can access our quality control protocols and view COA documents to verify peptide purity and sequence identity before beginning their trials.
Future Directions: KPV and The Mitochondria
Newer research is beginning to explore whether KPV’s reduction of oxidative stress has a downstream effect on mitochondrial health. While peptides like MOTS-c and SS-31 are the gold standards for direct mitochondrial targeting, the "anti-inflammatory axis" of KPV and BPC-157 may provide a permissive environment for mitochondrial recovery by lowering the metabolic burden of chronic inflammation.
Conclusion
The investigation of KPV and BPC-157 represents one of the most promising frontiers in epithelial research. By combining the profound anti-inflammatory signaling modulation of KPV with the robust regenerative and angiogenic properties of BPC-157, researchers can model complex healing processes that were previously difficult to address with single-agent therapies. As we continue to refine our understanding of peptide synthesis and in vitro modeling, this axis will likely serve as a cornerstone for future breakthroughs in metabolic and barrier-repair science.
For those interested in the underlying chemistry of these molecules, please visit our section on peptide synthesis to learn how we maintain the stability and bioactivity required for high-level research.
References
- 1. Sikiric P, et al. "Stable Gastric Pentadecapeptide BPC 157, Cortistatin, and Gastrointestinal Tract." Current Pharmaceutical Design, 2020.
- 2. Xiao B, et al. "PepT1-mediated tripeptide KPV uptake in intestinal epithelial cells and its anti-inflammatory effects." Digestive Diseases and Sciences, 2012.
- 3. Ceranowicz P, et al. "Therapeutic Effect of BPC 157 on Acute Pancreatitis." Molecules, 2020.
- 4. Dalmasso G, et al. "PepT1-mediated transport of the tripeptide KPV and its anti-inflammatory effects in intestinal epithelial cells." Gastroenterology, 2008.
- 5. Seiwerth S, et al. "BPC 157 and Blood Vessels." Frontiers in Pharmacology, 2021.
- 6. Scholzen TE, et al. "The Melanocortin Peptide KPV: A New Player in the Inflammation and Immune Response." Journal of Investigative Dermatology, 2010.
- 7. Chang CH, et al. "The promoting effect of pentadecapeptide BPC 157 on tendon-to-bone healing." Journal of Orthopaedic Research, 2011.
- 8. Land SC. "Control of intestinal epithelial permeability by melatonin and the alpha-melanocyte-stimulating hormone analog KPV." Journal of Pineal Research, 2013.
- 9. Brancati A, et al. "The role of the melanocortin system in inflammatory diseases." International Journal of Molecular Sciences, 2022.
- 10. Bitar MS, et al. "The Gastric Pentadecapeptide BPC 157 Corrects the Compromised Gastrointestinal and Cutaneous Healing." Journal of Physiology and Pharmacology, 2021.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.