The Amylin-Incretin Axis: Evaluating the Research Synergy of Cagrilintide and Semaglutide
This deep-dive analysis explores the synergistic "CagriSema" combination of Cagrilintide and Semaglutide, examining the molecular mechanisms of the Amylin-Incretin axis. We evaluate preclinical and Phase 2 data, structural engineering, and the physiological rationale behind this emerging dual-agonist approach in metabolic research.
The Next Frontier in Metabolic Research: Beyond Mono-Therapy
The landscape of metabolic research, particularly concerning obesity and type 2 diabetes mellitus, has undergone a radical transformation over the past decade. The era of small-molecule stimulants has largely been superseded by the age of peptide therapeutics, characterized by high specificity and potent mimicry of endogenous hormones. While the discovery and refinement of Glucagon-Like Peptide-1 (GLP-1) receptor agonists represented a watershed moment in scientific understanding of satiety signaling, contemporary research is now pivoting toward combinational approaches. The hypothesis driving this shift is that targeting multiple, non-overlapping neuroendocrine pathways can produce synergistic effects that far exceed the sum of their individual components.
Currently, the most prominent investigation in this domain involves the co-administration of Cagrilintide, a novel long-acting amylin analogue, and Semaglutide, the established GLP-1 receptor agonist. Often referred to in literature as "CagriSema," this combination represents a dual-hormonal approach that targets both the incretin axis and the amylin signaling pathway. For researchers working with peptide models, understanding the distinct yet complementary mechanisms of these two agents is crucial for experimental design and data interpretation.
This article provides a comprehensive evaluation of the physiological underpinnings, pharmacokinetic innovations, and synergistic potential of the amylin-incretin axis. We will dissect the molecular biology of both compounds, review pivotal preclinical and phase 2 trial data, and contextualize how this combination compares to other emerging multi-receptor agonists like Tirzepatide and Retatrutide.
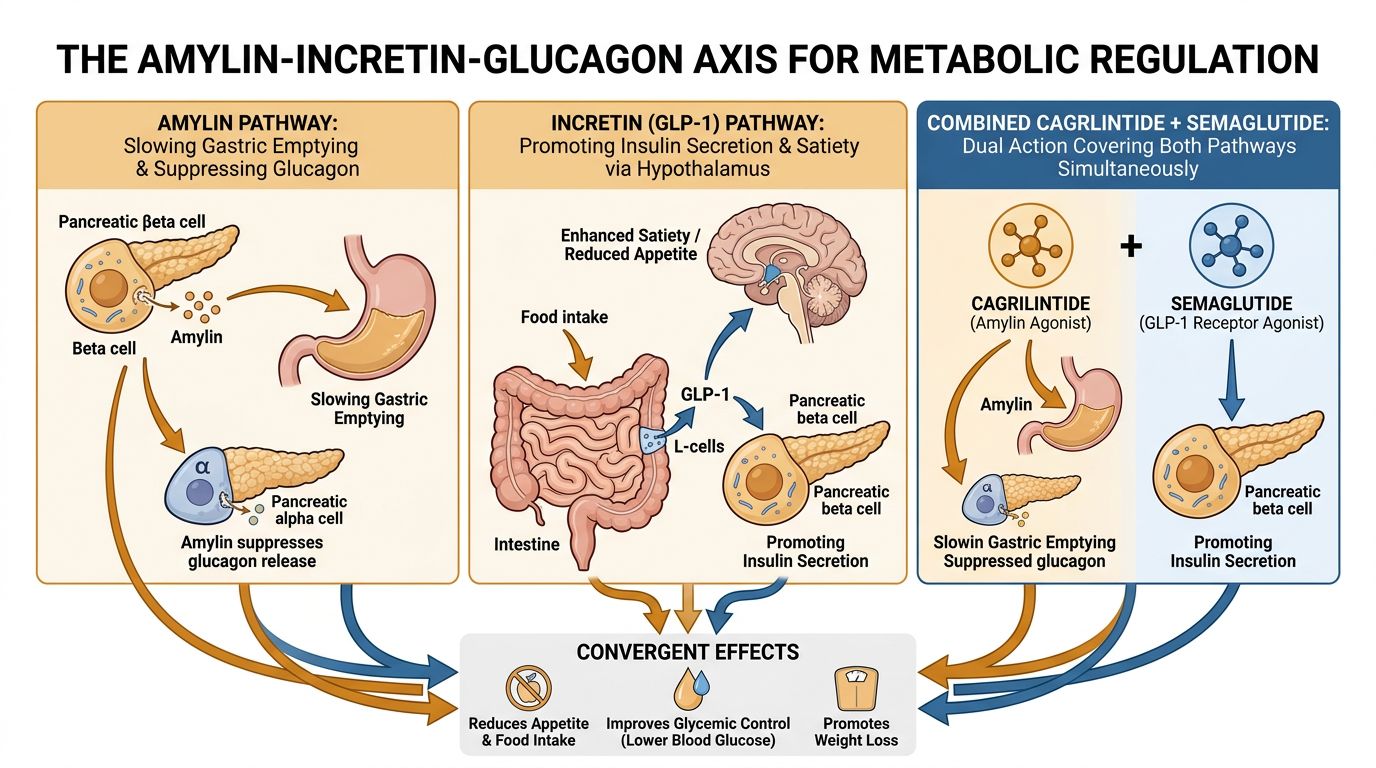 Cagrilintide plus Semaglutide dual action" style="max-width: 100%; height: auto; border-radius: 8px;" />
Cagrilintide plus Semaglutide dual action" style="max-width: 100%; height: auto; border-radius: 8px;" />The Incretin Arm: Semaglutide and GLP-1 Receptor Agonism
To understand the synergy, one must first isolate the components. Semaglutide is a modified analogue of human GLP-1, an incretin hormone primarily secreted by the L-cells of the distal ileum and colon in response to nutrient ingestion. Its effects are pleiotropic, influencing glucose homeostasis, gastric motility, and central appetite regulation.
Structural Modifications and Pharmacokinetics
Native GLP-1(7-37) has an extremely short half-life (approximately 1.5 to 2 minutes) due to rapid degradation by the enzyme dipeptidyl peptidase-4 (DPP-4) and renal clearance. For a research peptide to be viable in chronic administration studies, this limitation must be overcome. Semaglutide incorporates two critical structural modifications:
- Aib Substitution: A substitution of alpha-aminoisobutyric acid (Aib) at position 8 reinforces the peptide bond against DPP-4 cleavage.
- Acylation: The attachment of a C18 fatty diacid side chain via a hydrophilic spacer (gamma-glutamic acid and bis-aminouethoxyethoxy-acetyl conjugation). This fatty acid tail facilitates non-covalent binding to serum albumin.
Albumin binding serves a dual purpose: it protects the peptide from enzymatic degradation and minimizes renal filtration, extending the half-life to approximately 165 hours (in humans). This is what enables the once-weekly administration protocols seen in clinical literature.
Mechanism of Action: Central and Peripheral
Semaglutide exerts its weight-modulating effects primarily through the central nervous system (CNS). Although the GLP-1 receptor is expressed in the pancreas (enhancing glucose-dependent insulin secretion and suppressing glucagon), the profound weight loss observed in research models is attributed to CNS engagement. Fluorescently labeled Semaglutide has been shown to access specific brain regions, notably the hypothalamus and the brainstem.
Within the arcuate nucleus of the hypothalamus, GLP-1 agonists stimulate pro-opiomelanocortin (POMC) neurons—which induce satiety—and indirectly inhibit Neuropeptide Y (NPY) and Agouti-related peptide (AgRP) neurons—which drive hunger. Furthermore, GLP-1 engagement reduces the reward value of food via the mesolimbic dopamine system, potentially reducing cravings for palatable, high-calorie nutrition.
The Amylin Arm: Cagrilintide and the Challenge of Aggregation
While GLP-1 focuses on the "incretin" response, Amylin (Islet Amyloid Polypeptide or IAPP) represents a distinct neuroendocrine pathway. Co-secreted with insulin by pancreatic beta-cells in a 1:100 molar ratio, amylin functions as a synergistic partner to insulin in postprandial glucose control. However, its utility as a therapeutic or research agent was historically hampered by its physiochemical properties.
The Problem with Native Amylin
Human amylin is highly amyloidogenic. In aqueous solution, it rapidly aggregates to form insoluble fibrils (amyloid plaques), which are cytotoxic to beta-cells and render the peptide biologically inactive in therapeutic formulations. Early attempts to utilize amylin resulted in the development of Pramlintide, a non-aggregating analogue where three proline substitutions were introduced (mimicking rat amylin, which does not aggregate). However, Pramlintide has a very short half-life requiring multiple daily injections, limiting its widespread adoption in research protocols focusing on chronic weight management.
Enter Cagrilintide: Next-Generation Engineering
Cagrilintide represents a significant leap in peptide engineering. Like Semaglutide, it is designed for extended duration of action, but it specifically targets the amylin receptors (AMY). The structural logic mirrors the success of long-acting GLP-1s:
- Sequence Modification: Cagrilintide is an acylated analogue of human amylin. It contains specific amino acid substitutions to prevent fibril formation and aggregation, ensuring stability in solution.
- Protraction via Fatty Acid: Similar to Semaglutide, Cagrilintide is conjugated with a long-chain fatty diacid. This allows for albumin binding, protecting the molecule from renal clearance and enzymatic breakdown, effectively extending its half-life to support once-weekly administration in research subjects.
This stability allows researchers to study sustained amylin receptor agonism without the confounding variables of fluctuating plasma levels seen with short-acting analogues like Pramlintide.
Mechanism of Action: The Hindbrain Connection
Amylin’s mechanism of action is distinct from GLP-1. Amylin receptors are actually calcitonin receptors (CTR) heterodimerized with Receptor Activity-Modifying Proteins (RAMPs). The specific combination of CTR and RAMP determines the receptor subtype (AMY1, AMY2, AMY3).
Cagrilintide primarily acts on the Area Postrema (AP) and the Nucleus of the Solitary Tract (NTS) in the hindbrain. These regions are circumventricular organs, widely considered to be outside the Blood-Brain Barrier (BBB), making them highly accessible to circulating peptides. Upon binding, Cagrilintide triggers signaling cascades that signal "fullness" or satiation. Additionally, amylin agonism slows gastric emptying (a mechanism shared with GLP-1 but mediated differently) and suppresses postprandial glucagon secretion.
The Physiology of Synergy: Why Combine Them?
The core scientific question driving the "CagriSema" research is why a combination would be superior to simply increasing the dose of a single agent. The answer lies in the biological ceiling of mono-therapy and the non-overlapping distribution of receptors.
1. Distinct Neuronal Targets
While both peptides influence the CNS, they target different neuronal populations. GLP-1 receptors are dense in the hypothalamus (arcuate nucleus) and the mesolimbic system. Amylin receptors are highly concentrated in the hindbrain (AP/NTS). By stimulating both the "homeostatic" feeding centers (hypothalamus) and the "emergency/reflex" centers (hindbrain) simultaneously, the combination creates a more comprehensive blockade of orexigenic (hunger) signals.
2. Overcoming Counter-Regulatory Mechanisms
The body defends its adipose tissue vigorously. When extensive weight loss occurs via one pathway (e.g., GLP-1 agonism), counter-regulatory mechanisms often kick in to decrease energy expenditure or increase hunger drive via alternative pathways. By engaging the amylin pathway, which has a strong influence on satiation (the termination of a meal) rather than just satiety (the interval between meals), researchers hypothesize that the combination prevents the rigorous "plateau" effect seen in monotherapy studies.
3. Tolerability and Dosing
Dose-escalation of GLP-1 agonists is eventually limited by gastrointestinal side effects (nausea, emesis). There is a therapeutic window where efficacy is high but side effects are manageable. Adding an amylin agonist allows researchers to attack obesity from a second angle without pushing the GLP-1 dose into Intolerable ranges. Interestingly, preclinical data suggests amylin may enhance the weight-lowering efficacy of GLP-1 without proportionally increasing the nausea burden, although clinical data shows GI effects remain the primary adverse event for the combination.
Reviewing the Research: Preclinical and Clinical Efficacy
To evaluate the potential of the Cagrilintide and Semaglutide combination, we must examine the data generated from rigorously controlled trials. The results specifically highlight the additive—and often synergistic—nature of this pairing.
Preclinical Foundations
In diet-induced obese (DIO) rat models, co-administration of Cagrilintide and Semaglutide resulted in significantly greater weight loss than either agent alone. A pivotal study published in Molecular Metabolism (2022) demonstrated that the combination triggered a pronounced reduction in food intake and a shift in food preference, reducing the intake of palatable, high-fat chow. Furthermore, the study noted improvements in glycemic control that exceeded monotherapy, suggesting the suppression of glucagon by both agents acts in concert to normalize blood glucose.
Clinical Phase 2 Findings (The Frias Study)
The most significant dataset currently available comes from a Phase 2 trial published in The Lancet (Frias et al., 2023). This randomized, double-blind study involved patients with type 2 diabetes and overweight/obesity. The study compared the fixed-dose combination (CagriSema) against Semaglutide alone and Cagrilintide alone.
| Parameter | Semaglutide Mono (2.4mg) | Cagrilintide Mono (2.4mg) | CagriSema Combination (2.4mg/2.4mg) |
|---|---|---|---|
| Weight Loss (%) | ~5.1% | ~8.1% | ~15.6% |
| HbA1c Reduction | -1.5% | -0.9% | -2.2% |
| Primary Adverse Events | GI (Nausea/Diarrhea) | GI (Nausea) | GI (Comparable to Mono) |
Note: Data approximated from Phase 2 findings in T2D patients. Weight loss in non-diabetic obese cohorts is typically higher.
The data demonstrated a clear superiority of the combination. The weight loss achieved by CagriSema (15.6%) was roughly the sum of the individual components, validating the additive hypothesis. Importantly, the HbA1c reduction was also superior, suggesting that the dual mechanism offers better glycemic control, likely due to the potent suppression of postprandial glucose excursions mediated by amylin's effect on gastric emptying.
Comparative Potency: Versus Tirzepatide
When positioning CagriSema in the hierarchy of research peptides, the natural comparison is Tirzepatide (GLP-1/GIP co-agonist). While head-to-head trials are ongoing, indirect comparisons are valuable for researchers planning studies. Tirzepatide utilizes GIP (Glucose-dependent Insulinotropic Polypeptide) alongside GLP-1. GIP is generally adipogenic in isolation, but in the context of GLP-1 agonism, it appears to improve lipid handling and insulin sensitivity while reducing nausea.
CagriSema, by contrast, uses Amylin. Early projections suggest CagriSema may offer greater raw weight loss potential than Tirzepatide, potentially rivaling bariatric surgery results (20-25% reduction ranges). However, the side effect profile differs. The amylin component provides very strong satiety but carries a known risk of nausea, whereas GIP may mitigate some GLP-1 induced nausea. Researchers choosing between these substrates must weigh the goal of maximal weight reduction (CagriSema) against metabolic modulation and lipid handling (Tirzepatide).
Peptide Engineering and Quality Considerations
For independent researchers utilizing these peptides, understanding the complexity of their synthesis is paramount. Both Cagrilintide and Semaglutide are large, complex molecules requiring precise solid-phase peptide synthesis (SPPS) and subsequent modification (acylation).
Solubility and Stability
The addition of fatty acid chains renders these peptides hydrophobic in certain domains, creating solubility challenges relative to simple linear peptides like GHK-Cu or BPC-157. Proper reconstitution buffers (bacteriostatic water is standard, but pH plays a role) and storage conditions are critical. In a research setting, ensuring the peptide has not aggregated—specifically for the amylin component, despite the engineering—is a key validity check.
Purity Verification
The complexity of the acylation process introduces the possibility of specific impurities, such as incorrect fatty acid attachment sites or incomplete conjugation. High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) are non-negotiable for verifying the identity and purity of these compounds. Researchers are advised to review COA Documents meticulously to ensure the absence of trifluoroacetic acid (TFA) salts or deletion sequences that could alter experimental outcomes. For more on how these peptides are manufactured and verified, visit our peptide synthesis overview.
Safety Profile and Research Monitoring
While peptide therapeutics are generally safer than small molecule stimulants due to their specificity, the potency of the Cagrilintide/Semaglutide combination mandates careful safety monitoring in any study protocol.
Gastrointestinal Impact
The most pervasive side effects are gastrointestinal. Nausea, vomiting, and diarrhea are dose-dependent. In Phase 2 trials, discontinuation rates due to adverse events were higher in the CagriSema group compared to Semaglutide alone. Research protocols should implement dose-titration strategies (gradually increasing concentrations over 4-16 weeks) to mitigate these effects and prevent subject attrition.
Pancreatic and Biliary Safety
As with all incretin-based therapies, markers for pancreatitis (amylase/lipase) and gallbladder function should be monitored. While a causal link remains a subject of debate in the wider medical community, research ethics dictate vigilant observation of these parameters. The amylin component does not historically carry a pancreatitis risk, but the slowing of gallbladder motility (cholelithiasis risk) is a known class effect of rapid weight loss induced by GLP-1s.
Cardiovascular Considerations
Semaglutide has demonstrated cardioprotective benefits (as seen in the SELECT trial). It is hypothesized that Cagrilintide will not negate these benefits, and may enhance them via substantial weight reduction. However, researchers monitoring heart rate should note that GLP-1 agonists consistently cause a slight elevation in resting heart rate (2-5 bpm). The implications of this in the context of amylin co-administration are currently under investigation.
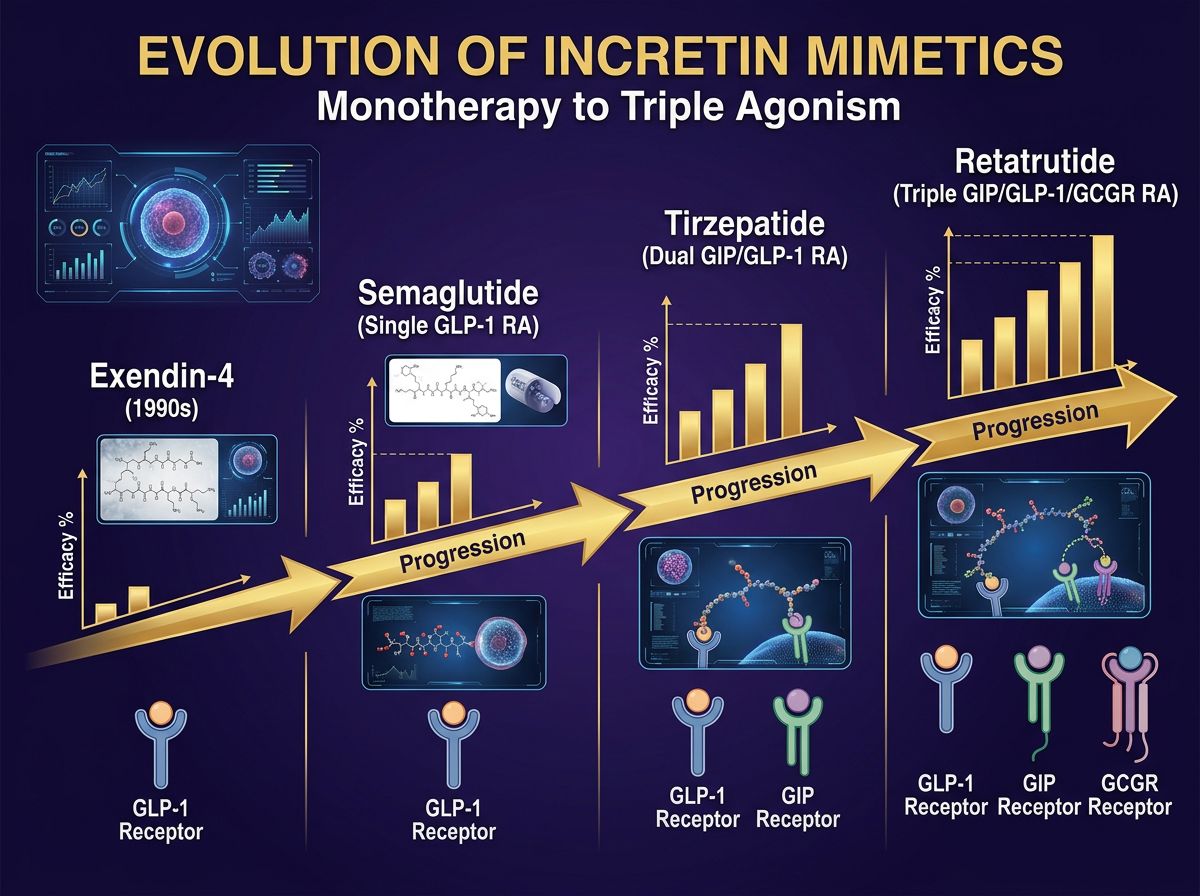
Future Directions: The Rise of Triple Agonists and Beyond
The investigation of Cagrilintide and Semaglutide is merely the current apex of a rapidly evolving field. As researchers validate the efficacy of the Amylin-Incretin axis, the industry is already looking toward triple-agonism.
Compounds like Retatrutide (GLP-1/GIP/Glucagon tri-agonist) are entering the fray. Retatrutide introduces the glucagon receptor (GCGR) into the mix, which increases energy expenditure (thermogenesis) via brown adipose tissue activation and liver lipid catabolism. This contrasts with CagriSema, which is primarily an "intake restriction" mechanism.
This creates a fascinating divergence in research possibilities:
- Pathway A (CagriSema): Maximizing satiety and suppression of intake via Amylin + GLP-1. Ideal for models investigating pure caloric restriction and gastric emptying.
- Pathway B (Retatrutide/Mazdutide): modulating energy expenditure and lipid metabolism via Glucagon + GLP-1 (+ GIP). Ideal for models of fatty liver disease (MASLD) and metabolic rate modulation.
Additionally, other amylin analogues are being screened, and combinations involving Mazdutide (GLP-1/Glucagon dual agonist) are being tested against CagriSema to determine whether burning more calories or eating fewer calories leads to better long-term tissue health.
Conclusion
The co-administration of Cagrilintide and Semaglutide represents a sophisticated evolution in peptide-based metabolic research. By bridging the Incretin and Amylin axes, this combination offers a dual-pronged attack on adiposity that monotherapies cannot replicate. The synergy observed in both preclinical rat models and Phase 2 human trials suggests that targeting the hindbrain (via amylin) alongside the hypothalamus (via GLP-1) creates a robust blockade against compensatory hunger signals.
For researchers, the "CagriSema" combination serves as a prime investigation tool for high-efficacy weight loss, surpassing the capabilities of first-generation GLP-1 agonists. However, the rigor of research protocols must account for the complex solubility of these acylated peptides and the significant gastrointestinal signaling they induce. As Alpha Carbon Labs continues to support the scientific community with high-purity research peptides, we anticipate that the data generated from this specific combination will pave the way for a new standard in obesity, diabetes, and metabolic syndrome research.
For further technical specifications on these compounds and to verify batch purity, please refer to our quality control protocols.
References
- Frias, J. P., et al. (2023). Efficacy and safety of co-administered cagrilintide and semaglutide (CagriSema) in people with type 2 diabetes: a multicentre, double-blind, randomised, phase 2 trial. The Lancet, 402(10403), 720-730. https://doi.org/10.1016/S0140-6736(23)01163-7
- Enebo, L. B., et al. (2021). Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide in diverse weight management populations. The Lancet, 397(10286), 1715-1724. https://doi.org/10.1016/S0140-6736(21)00845-X
- Kruse, T., et al. (2021). The amylin analogue cagrilintide reduces food intake and body weight in rats via the amylin receptor in the hindbrain. British Journal of Pharmacology, 178(20), 4162-4176. https://doi.org/10.1111/bph.15609
- Raun, K., et al. (2022). Cagrilintide, a long-acting amylin analogue, acts synergistically with semaglutide to reduce body weight in diet-induced obese rats. Molecular Metabolism, 60, 101490. https://doi.org/10.1016/j.molmet.2022.101490
- Lau, J., et al. (2015). Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry, 58(18), 7370-7380. https://doi.org/10.1021/acs.jmedchem.5b00726
- Boyle, C. N., et al. (2018). Role of amylin in the control of energy homeostasis. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 315(3), R499-R511. https://doi.org/10.1152/ajpregu.00096.2018
- Knudsen, L. B., & Lau, J. (2019). The Discovery and Development of Liraglutide and Semaglutide. Frontiers in Endocrinology, 10, 155. https://doi.org/10.3389/fendo.2019.00155
- DePaoli, A. M. (2024). The evolution of amylin agonists for the treatment of obesity. Peptides, 172, 171132. https://doi.org/10.1016/j.peptides.2023.171132
- Soderstrom, K., et al. (2020). Pharmacology of the GIP/GLP-1 receptor co-agonist tirzepatide. Journal of Diabetes and its Complications, 34(12), 107722. https://doi.org/10.1016/j.jdiacomp.2020.107722
- Gault, V. A., & Holscher, C. (2018). GLP-1 and amylin signalling strategies related to diabetes and Alzheimer's disease. Neuropeptides, 68, 64-70. https://doi.org/10.1016/j.npep.2018.02.001
References
- 1. Frias, J. P., et al. (2023). Efficacy and safety of co-administered cagrilintide and semaglutide (CagriSema) in people with type 2 diabetes: a multicentre, double-blind, randomised, phase 2 trial. The Lancet, 402(10403), 720-730.
- 2. Enebo, L. B., et al. (2021). Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide in diverse weight management populations. The Lancet, 397(10286), 1715-1724.
- 3. Lau, J., et al. (2015). Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry, 58(18), 7370-7380.
- 4. Kruse, T., et al. (2021). The amylin analogue cagrilintide reduces food intake and body weight in rats via the amylin receptor in the hindbrain. British Journal of Pharmacology, 178(20), 4162-4176.
- 5. Boyle, C. N., et al. (2018). Role of amylin in the control of energy homeostasis. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 315(3), R499-R511.
- 6. DePaoli, A. M. (2024). The evolution of amylin agonists for the treatment of obesity. Peptides, 172, 171132.
- 7. Knudsen, L. B., & Lau, J. (2019). The Discovery and Development of Liraglutide and Semaglutide. Frontiers in Endocrinology, 10, 155.
- 8. Gault, V. A., & Holscher, C. (2018). GLP-1 and amylin signalling strategies related to diabetes and Alzheimer's disease. Neuropeptides, 68, 64-70.
- 9. Raun, K., et al. (2022). Cagrilintide, a long-acting amylin analogue, acts synergistically with semaglutide to reduce body weight in diet-induced obese rats. Molecular Metabolism, 60, 101490.
- 10. Soderstrom, K., et al. (2020). Pharmacology of the GIP/GLP-1 receptor co-agonist tirzepatide. Journal of Diabetes and its Complications, 34(12), 107722.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.