SLU-PP-332: ERR Agonist Research
Explore SLU-PP-332's mechanism as an ERR agonist for energy expenditure, fatty acid oxidation, and exercise mimetic research.
Introduction: Estrogen-Related Receptors and Metabolic Regulation
SLU-PP-332 is a potent, selective pan-estrogen related receptor (ERR) agonist that has emerged as a valuable research tool for studying metabolic regulation and energy homeostasis. Developed at Saint Louis University (hence the "SLU" designation), this small molecule compound selectively activates all three members of the ERR family—ERRα, ERRβ, and ERRγ—with high potency and selectivity over other nuclear receptors.
The ERR family represents a group of orphan nuclear receptors that, despite their name, do not bind estrogen but share some DNA target sequences with classical estrogen receptors. These transcription factors play fundamental roles in regulating cellular energy metabolism, serving as master regulators of genes involved in oxidative phosphorylation, fatty acid oxidation, and mitochondrial biogenesis.
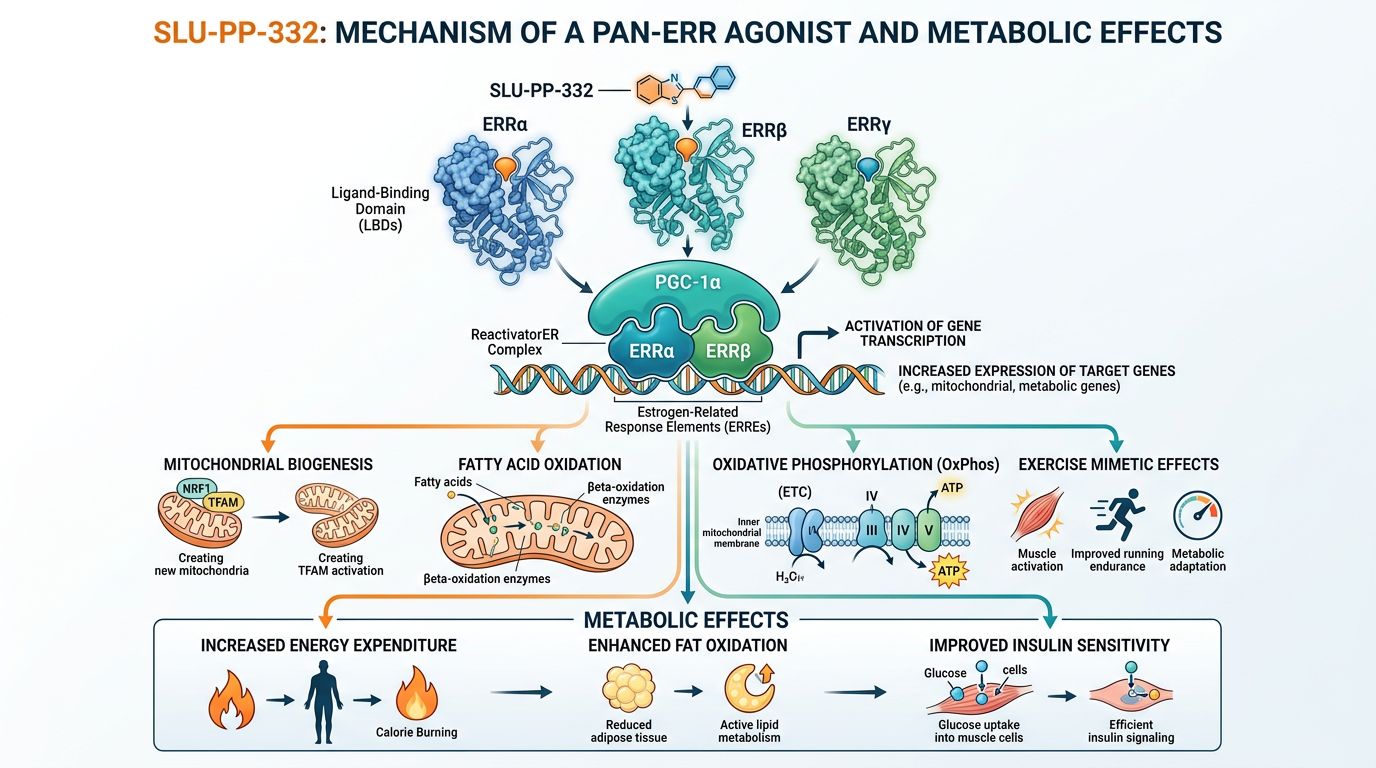
Molecular Structure and Receptor Selectivity
SLU-PP-332, chemically known as (E)-4-hydroxy-N'-(naphthalen-2-ylmethylene)benzohydrazide, activates ERR isoforms with distinct potencies. Research has established EC50 values of approximately 98 nM for ERRα, 230 nM for ERRβ, and 430 nM for ERRγ, making it among the most potent pan-ERR agonists available for research purposes.
This selectivity profile is significant because each ERR isoform has distinct tissue distribution and functional characteristics. ERRα is highly expressed in tissues with high energy demands—heart, skeletal muscle, brown adipose tissue, and kidney—where it coordinates expression of metabolic gene networks essential for oxidative capacity. ERRβ and ERRγ have more restricted expression patterns but contribute to tissue-specific metabolic regulation.
Importantly, SLU-PP-332 demonstrates minimal activity at estrogen receptors (ERα and ERβ) and other nuclear receptors, allowing researchers to isolate ERR-specific effects without confounding activity at related receptors. This selectivity distinguishes it from earlier compounds that exhibited significant cross-reactivity.
Mechanism of Action: Nuclear Receptor Activation
ERRs are constitutively active nuclear receptors, meaning they can regulate gene expression even without a bound ligand. However, their transcriptional activity is significantly enhanced by interaction with coactivators, particularly the peroxisome proliferator-activated receptor gamma coactivator-1 alpha and beta (PGC-1α and PGC-1β).
SLU-PP-332 binds to the ligand-binding domain (LBD) of ERRs and stabilizes the receptor in an active conformation that enhances coactivator recruitment. This leads to increased binding of ERRs to ERR response elements (ERREs) in the promoters and enhancers of target genes, promoting transcription of metabolic gene programs.
Key target genes of ERR activation include those encoding components of the electron transport chain (such as cytochrome c and ATP synthase subunits), fatty acid oxidation enzymes (including medium-chain acyl-CoA dehydrogenase and carnitine palmitoyltransferase-1), and transcription factors that amplify the metabolic signal. This comprehensive activation of metabolic gene expression results in enhanced mitochondrial function and increased oxidative capacity.
Metabolic Effects: Energy Expenditure and Fat Oxidation
Research with SLU-PP-332 has demonstrated remarkable effects on whole-body metabolism in preclinical models. Treatment of mice with metabolic syndrome produced several notable outcomes without changes in food intake—a critical observation suggesting effects on energy expenditure rather than appetite suppression.
Studies showed that SLU-PP-332 administration increased energy expenditure, as measured by indirect calorimetry. Animals treated with the compound demonstrated elevated oxygen consumption and carbon dioxide production, indicating enhanced oxidative metabolism. This increase in energy expenditure was accompanied by a shift in substrate utilization toward fatty acid oxidation, evidenced by decreased respiratory exchange ratio.
Perhaps most significantly, treated animals showed decreased fat mass despite maintaining similar food intake as controls. This reduction in adiposity occurred preferentially without significant changes in lean mass, suggesting that SLU-PP-332 promotes fat utilization while preserving or enhancing muscle oxidative capacity. These findings align with the known role of ERRs in regulating lipid metabolism and energy balance.
Insulin Sensitivity and Glucose Homeostasis
Beyond effects on body composition, SLU-PP-332 treatment improved insulin sensitivity in metabolic syndrome models. Glucose tolerance tests revealed enhanced glucose disposal in treated animals, with corresponding improvements in insulin responsiveness. These metabolic improvements are likely secondary to reduced adiposity and enhanced muscle oxidative capacity.
Skeletal muscle, which accounts for the majority of insulin-stimulated glucose disposal, shows particularly robust ERR expression. By enhancing muscle oxidative capacity and fatty acid utilization, ERR activation may reduce lipid accumulation within muscle cells—a phenomenon linked to insulin resistance—thereby improving glucose metabolism. Additionally, the enhanced mitochondrial function may improve the efficiency of glucose oxidation once glucose enters the cell.
These findings have implications for understanding metabolic flexibility—the ability of tissues to switch between fuel sources based on availability and demand. ERR activation appears to enhance this metabolic flexibility, potentially explaining the broad metabolic improvements observed with SLU-PP-332 treatment.
Mitochondrial Biogenesis and Function
A central aspect of ERR biology is the regulation of mitochondrial biogenesis—the process by which cells increase their mitochondrial content and capacity. ERRs work in concert with PGC-1α, often called the "master regulator" of mitochondrial biogenesis, to coordinate the expression of nuclear and mitochondrial genes encoding components of the oxidative phosphorylation machinery.
SLU-PP-332 treatment increases mitochondrial respiration in vitro, as demonstrated in studies using cultured cells and isolated mitochondria. This enhanced respiratory capacity reflects increased expression of electron transport chain components and enzymes involved in substrate oxidation. The result is greater ATP-generating capacity per mitochondrion, as well as potential increases in mitochondrial number.
These effects have implications for understanding and potentially addressing conditions characterized by mitochondrial dysfunction. From primary mitochondrial diseases to the mitochondrial decline associated with aging, the ability to pharmacologically enhance mitochondrial function through ERR activation represents an intriguing research avenue.
Exercise Mimetic Research: Replicating Training Adaptations
The effects of SLU-PP-332 on oxidative metabolism and endurance capacity have generated interest in ERR agonists as potential "exercise mimetics"—compounds that could reproduce some molecular and metabolic benefits of physical exercise without physical activity. The transcriptional programs induced by ERR activation substantially overlap with those activated by endurance exercise training.
Exercise activates ERRs through multiple mechanisms, including increased PGC-1α expression and post-translational modifications that enhance ERR activity. The resulting adaptations—increased mitochondrial content, enhanced fatty acid oxidation, improved insulin sensitivity, and greater endurance capacity—are hallmarks of the trained state. By directly activating ERRs, SLU-PP-332 may bypass the need for exercise-induced signaling and directly promote these metabolic adaptations.
While exercise has far more effects than any single pathway modulator (including cardiovascular, neuromuscular, and skeletal adaptations), ERR activation does appear to replicate aspects of exercise training at the molecular and metabolic level. This has stimulated research into potential applications for individuals unable to exercise due to disability, illness, or other factors.
Tissue-Specific Effects: Heart, Muscle, and Brown Fat
ERRs show differential expression across tissues, and SLU-PP-332's effects vary accordingly. In the heart, ERRα is essential for maintaining cardiac energy metabolism, regulating genes involved in fatty acid oxidation (the heart's preferred fuel under normal conditions) and mitochondrial function. ERR activation may support cardiac energetics under stress conditions, though the specific effects require careful investigation given the heart's unique metabolic demands.
Skeletal muscle represents another key target tissue for ERR agonists. ERR activation promotes a fiber-type shift toward a more oxidative phenotype, characterized by increased expression of slow-twitch fiber markers, greater mitochondrial density, and enhanced fatigue resistance. These adaptations mirror those observed with endurance training.
Brown and beige adipose tissue, specialized for thermogenic energy expenditure, also express ERRs. These tissues contain high mitochondrial content and express uncoupling protein 1 (UCP1), which dissipates the proton gradient to generate heat rather than ATP. ERR activation may enhance thermogenic capacity, contributing to increased energy expenditure and reduced adiposity.
Comparison with Other Metabolic Modulators
SLU-PP-332 represents one approach to metabolic modulation through nuclear receptor activation. Other compounds with exercise-mimetic or metabolic-enhancing properties include AICAR (an AMPK activator), GW501516 (a PPARδ agonist), and various sirtuin activators. Each targets different but interconnected metabolic pathways.
The ERR pathway intersects with these other metabolic regulators through shared coactivators and overlapping target genes. PGC-1α, for example, serves as a coactivator not only for ERRs but also for PPARs and other nuclear receptors. AMPK activation increases PGC-1α expression and activity, indirectly enhancing ERR signaling. Understanding these interconnections is essential for developing optimal metabolic interventions.
SLU-PP-332's selectivity for ERRs allows researchers to isolate the specific contribution of this pathway to metabolic regulation, providing insights that complement studies using broader metabolic modulators.
Research Applications and Experimental Considerations
SLU-PP-332 serves as a valuable tool compound for investigating ERR biology across multiple research contexts. Primary applications include studies of metabolic syndrome and obesity mechanisms, fatty acid oxidation and lipid metabolism regulation, mitochondrial biogenesis and function, insulin sensitivity and glucose homeostasis, skeletal muscle metabolism and exercise physiology, cardiac energy metabolism, and brown and beige adipose tissue thermogenesis.
For in vitro studies, the compound is typically dissolved in DMSO and applied at micromolar concentrations. For in vivo research, various administration routes and dosing schedules have been employed, with careful attention to vehicle selection and pharmacokinetic considerations. Researchers should consult published protocols and optimize conditions for their specific experimental systems.
Safety Considerations in Research Use
SLU-PP-332 is a research compound that has been primarily characterized in preclinical studies, with limited data on human safety. As a pan-ERR agonist affecting metabolic gene expression across multiple tissues, several theoretical considerations are relevant for research use.
ERRs are expressed in tissues beyond those primarily involved in energy metabolism, including reproductive tissues, bone, and the brain. Systemic ERR modulation could have effects on these tissues that are not fully characterized. Additionally, while SLU-PP-332 is selective for ERRs over estrogen receptors in biochemical assays, the functional consequences of any residual cross-reactivity require consideration in experimental design.
Preclinical studies have generally not reported overt toxicity at efficacious doses. However, long-term effects of sustained ERR activation have not been fully evaluated. Standard precautions for handling small molecule research compounds apply.
Future Directions in ERR Research
Research with SLU-PP-332 and related ERR modulators continues to expand our understanding of metabolic regulation. Key questions for future investigation include identifying optimal dosing strategies for different research applications, characterizing tissue-specific effects and potential off-target activities, exploring combination approaches with other metabolic modulators, and understanding the long-term effects of ERR activation on various physiological systems.
The development of isoform-selective ERR modulators represents another research frontier, as selective activation of ERRα, ERRβ, or ERRγ could provide more targeted effects with potentially improved safety profiles. Such compounds would also help delineate the specific contributions of each isoform to metabolic regulation.
Conclusion
SLU-PP-332 represents a significant advance in research tools for studying metabolic regulation through the ERR pathway. By selectively activating the ERR family of orphan nuclear receptors, this compound enables investigation of fundamental questions about energy homeostasis, mitochondrial function, and the molecular basis of exercise adaptations. Research findings demonstrating enhanced energy expenditure, increased fatty acid oxidation, reduced adiposity, and improved insulin sensitivity highlight the central role of ERRs in metabolic regulation and suggest potential therapeutic applications for metabolic diseases. As an exercise mimetic research tool, SLU-PP-332 provides insights into how pharmacological approaches might replicate some benefits of physical training, with implications for individuals unable to exercise. Continued research will further elucidate the potential and limitations of ERR activation for metabolic research applications.
References
- 1. Willy, P. J., et al. (2004). Regulation of PPARγ coactivator 1α (PGC-1α) signaling by an estrogen-related receptor α (ERRα) ligand. PNAS, 101(24), 8912-8917.
- 2. Rangwala, S. M., et al. (2010). Estrogen-related receptor gamma is a key regulator of muscle mitochondrial activity and oxidative capacity. Journal of Biological Chemistry, 285(29), 22619-22629.
- 3. Giguère, V. (2008). Transcriptional control of energy homeostasis by the estrogen-related receptors. Endocrine Reviews, 29(6), 677-696.
- 4. Huss, J. M., et al. (2007). Estrogen-related receptor α directs peroxisome proliferator-activated receptor α signaling in the transcriptional control of energy metabolism in cardiac and skeletal muscle. Molecular and Cellular Biology, 27(14), 5406-5417.
- 5. Narkar, V. A., et al. (2011). Exercise and PGC-1α-independent synchronization of type I muscle metabolism and vasculature by ERRγ. Cell Metabolism, 13(3), 283-293.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.