Pharmacokinetic Divergence: Analyzing Bioavailability Variations in Semaglutide Delivery Models
An in-depth scientific analysis of the pharmacokinetic differences between oral and subcutaneous Semaglutide, focusing on bioavailability, SNAC technology, and receptor affinity.
Pharmacokinetic Divergence: Analyzing Bioavailability Variations in Semaglutide Delivery Models
The evolution of Glucagon-Like Peptide-1 (GLP-1) receptor agonists has shifted the landscape of metabolic research. Central to this evolution is Semaglutide, a potent analog that has demonstrated remarkable stability and receptor affinity. However, the divergence in its pharmacokinetic (PK) profile when delivered via subcutaneous (SC) versus oral routes presents a complex subject for laboratory analysis. Researchers must understand how delivery modality influences absolute bioavailability, metabolic velocity, and the steady-state concentration required to elicit specific biological responses.
Structural Biochemistry of Semaglutide
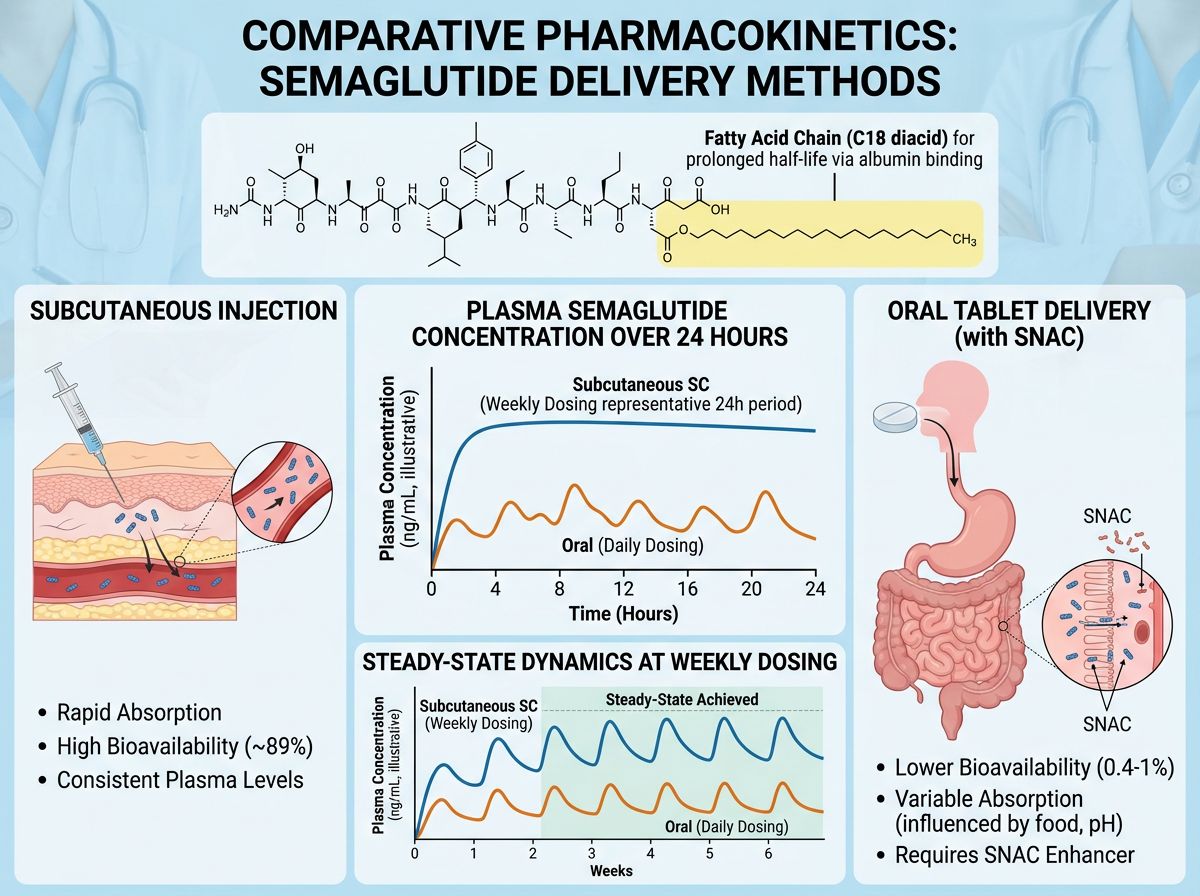
To understand the delivery divergence, one must first look at the molecular architecture of the peptide. Semaglutide is a 31-amino acid peptide that shares 94% structural homology with native human GLP-1. Three critical modifications contribute to its long half-life:
- Substitution of Alanine with alpha-aminoisobutyric acid (Aib) at position 8, which provides resistance to Dipeptidyl Peptidase-4 (DPP-4) degradation.
- The attachment of a C18 fatty diacid chain via a linker at position 26.
- Substitution of Lysine for Arginine at position 34 to ensure singular acylation.
The Challenge of Oral Peptide Delivery
Peptide research has historically been restricted by the "biologic barrier" of the gastrointestinal (GI) tract. Proteolytic enzymes and the acidic environment of the stomach typically degrade peptides before they can reach systemic circulation. In oral Semaglutide models, the peptide is co-formulated with an absorption enhancer: Sodium N-(8-[2-hydroxybenzoyl] amino) caprylate (SNAC).
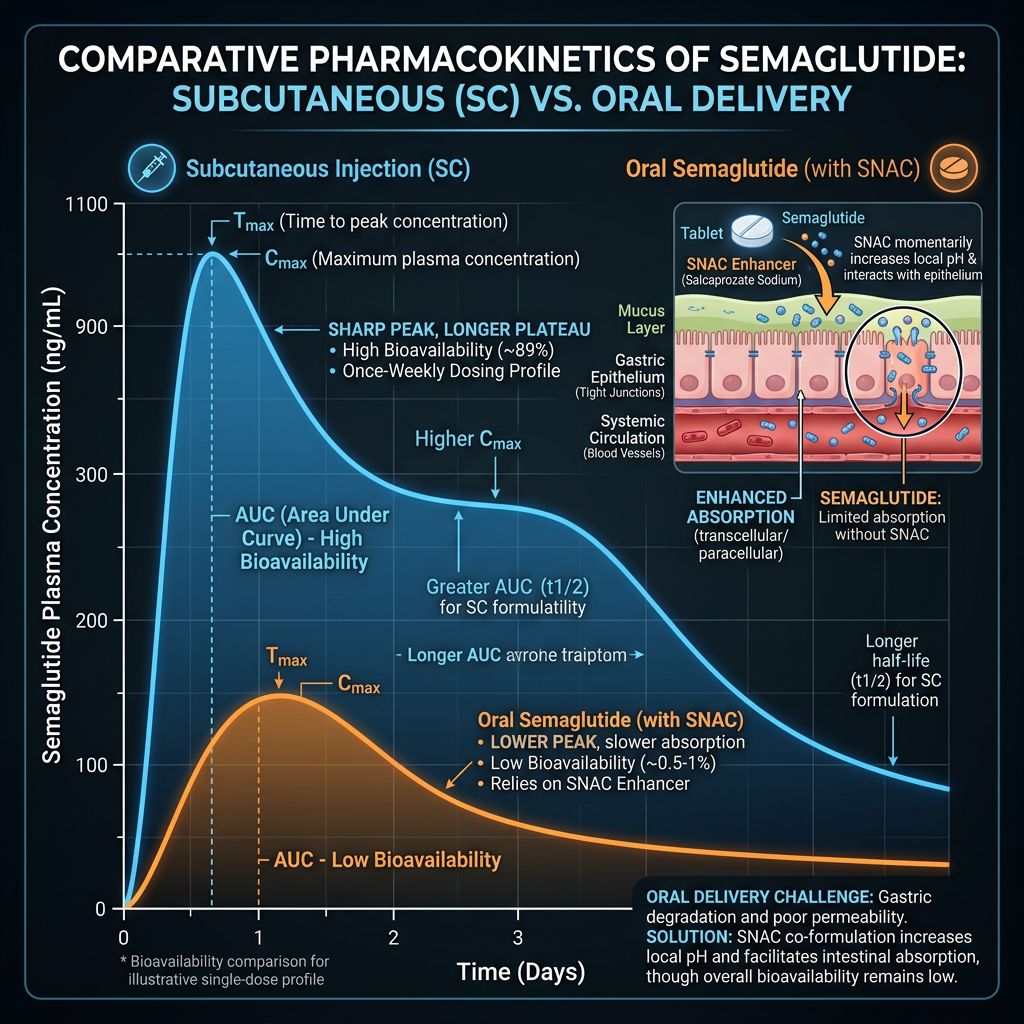
SNAC functions as a permeation enhancer that locally neutralizes the pH of the stomach, protecting the peptide from pepsin-mediated degradation. Simultaneously, it promotes the transcellular transport of Semaglutide across the gastric epithelium. In a research setting, the efficiency of this process is markedly lower than parenteral administration. While subcutaneous Semaglutide boasts an absolute bioavailability of approximately 89%, the oral variant typically exhibits a bioavailability of 0.4% to 1.2%.
Comparison Table: Subcutaneous vs. Oral Pharmacokinetics
| Parameter | Subcutaneous (SC) Delivery | Oral Delivery (+SNAC) |
|---|---|---|
| Absolute Bioavailability | ~89% | ~0.4 - 1.2% |
| Time to Maximum Concentration (Tmax) | 24–36 Hours | 1 Hour |
| Metabolic Half-Life | ~165 Hours | ~160 Hours |
| Absorption Site | Adipose/Interstitial Tissue | Gastric Mucosa (Stomach) |
| Affinity for GLP-1R | High (Direct Systemic) | High (Post-Portal Passage) |
Metabolic Velocity and Steady-State Dynamics
Metabolic velocity refers to the rate at which a compound reaches its target receptors and is subsequently cleared. In laboratory models using subcutaneous Semaglutide, the peptide reaches a steady state over several weeks due to its long half-life and slowed absorption from the injection site. This creates a "repository effect," where the interstitial fluid acts as a buffer, releasing the peptide into the blood at a controlled rate.
Conversely, oral delivery models show a rapid Tmax, often within one hour of administration. However, because the total amount absorbed is so small compared to the dose administered (requiring mg-scale oral doses vs mcg-scale SC doses), the systemic fluctuations are more sensitive to experimental variables. For instance, the presence of food or variations in gastric fluid volume can significantly alter the metabolic velocity of oral Semaglutide in animal models, a factor that is largely bypassed in subcutaneous research.
Receptor Affinity and Downstream Signaling
Once Semaglutide reaches the systemic circulation, its affinity for the GLP-1 receptor (GLP-1R) is independent of the delivery route. This receptor is a G protein-coupled receptor found in high concentrations in the pancreatic islets, hypothalamus, and enteric nervous system. The primary research focus often involves the cyclic AMP (cAMP) pathway, where Semaglutide binding triggers insulin secretion in a glucose-dependent manner.
However, researchers investigating neuroprotective or cardiovascular effects may prefer models that offer higher peak concentrations. Subcutaneous delivery provides a higher area-under-the-curve (AUC) for a given dose, potentially leading to more consistent receptor saturation in peripheral tissues compared to the oral route, where significant "first-pass" metabolism or hepatic clearance may occur before the peptide reaches the brain or heart.
Synergistic Research Models
In many research protocols, GLP-1 analogs like Semaglutide or Tirzepatide are studied alongside other metabolic peptides. To understand systemic energy expenditure, researchers may combine GLP-1Rs with mitochondrial modulators like MOTS-c or metabolic enhancers such as 5-amino-1mq. The delivery model of the GLP-1 analog can influence the experimental design for these combinations. For example, the consistent levels provided by subcutaneous Semaglutide often make it a more stable baseline for measuring the acute effects of secondary compounds.
Advanced Delivery and Future Implications
The "Second Generation" of GLP-1 research has expanded into dual and triple agonists. Recent studies on Retatrutide (a triple agonist of GLP-1, GIP, and Glucagon receptors) and Mazdutide suggest that multi-receptor targets follow similar PK rules to Semaglutide. The quest to improve oral bioavailability continues, with research focusing on nanotechnology-based carriers and enteric-coated formulations that protect the peptide further into the small intestine.
For research involving rapid weight-loss modeling and adipocyte signaling, compounds like Cagrilintide or Survodutide are currently being scrutinized in comparison to Semaglutide’s established PK profile to determine which delivery systems optimize the "incretin effect" without inducing excessive GI distress in laboratory subjects.
Quality Control in Peptide Research
Regardless of the delivery model—whether researching BPC-157 for tissue repair or Semaglutide for metabolic health—the integrity of the peptide sequence is paramount. Impurities or truncated sequences can drastically alter the PK profile and receptor affinity. Alpha Carbon Labs ensures that every batch meets rigorous standards through high-performance liquid chromatography (HPLC) and mass spectrometry. Researchers can access these findings via our COA documents to ensure their metabolic calculations are based on high-purity materials.
Conclusion
The choice between oral and subcutaneous delivery in Semaglutide research is not merely a matter of convenience but a decision that impacts the fundamental pharmacokinetics of the study. While oral delivery provides a unique model for gastric absorption and SNAC interactions, subcutaneous delivery remains the gold standard for high-bioavailability, consistent steady-state research. Understanding the divergence in metabolic velocity and absorption mechanics allows researchers to better interpret the resulting biological data and refine their metabolic models.
References
- 1. Knudsen LB, Lau J. The Discovery and Development of Semaglutide. Front Endocrinol (Lausanne). 2019;10:155.
- 2. Buckley ST, et al. Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist. Sci Transl Med. 2018;10(456).
- 3. Lau J, et al. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. J Med Chem. 2015;58(18):7370-80.
- 4. Granhall C, et al. Pharmacokinetics, Safety and Tolerability of Oral Semaglutide in Subjects with Renal Impairment. Clin Pharmacokinet. 2018;57(12):1571-1580.
- 5. Kapitza C, et al. Effects of semaglutide on beta cell function and glycemic control in patients with type 2 diabetes. Diabetologia. 2017;60(8):1390-1399.
- 6. Drucker DJ. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metab. 2018;27(4):740-756.
- 7. Al-Sabah S, et al. Structure-activity relationships of the glucagon-like peptide-1 receptor. Br J Pharmacol. 2015;172(20):4944-57.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.