Inhibiting the Myostatin Pathway: Evaluating ACE-031 vs. TB-500 in Myocellular Hypertrophy Studies
A detailed scientific comparison of ACE-031 and TB-500, exploring how one inhibits myostatin to unlock genetic growth potential while the other facilitates cellular repair and structural integrity.
Introduction: The Dual Frontiers of Muscle Science
In the realm of regenerative biology and musculoskeletal research, two distinct paradigms often dominate the conversation: the stimulation of growth beyond homeostatic limits, and the rapid repair of tissue to support that growth. These paradigms are represented by two of the most heavily investigated peptides in current literature: ACE-031 and TB-500. While both are frequently discussed in the context of performance and hypertrophy, their mechanisms of action are fundamentally different, operating on disparate biological axes.
Research into myocellular hypertrophy—the enlargement of muscle cells—has traditionally focused on anabolic signaling pathways involving testosterone, IGF-1, and mechanical tension. However, a significant breakthrough occurred with the identification of negative regulators of muscle mass, specifically the myostatin pathway. ACE-031 represents a sophisticated biotechnological intervention designed to inhibit these negative regulators, effectively removing the genetic "brakes" on muscle growth.
Conversely, the structural integrity of muscle tissue, particularly during phases of rapid expansion or injury, relies on cytoskeletal dynamics and angiogenesis. TB-500, a synthetic derivative of the naturally occurring protein Thymosin Beta-4, operates as a master regulator of actin sequestration and cell migration. It does not directly force hypertrophy via the same pathways as ACE-031; rather, it facilitates the structural environment necessary for tissue survival and recovery.
This article provides a comprehensive, comparative analysis of these two peptides. We will evaluate the molecular biology of ACE-031’s "decoy receptor" capability against the structural engineering properties of TB-500, exploring how they function in isolation and how their contrasting mechanisms illuminate the complexities of muscle physiology.
The Molecular Barrier: Understanding the Myostatin Pathway
To fully appreciate the function of ACE-031, one must first understand the biological system it disrupts: the Transforming Growth Factor-beta (TGF-β) superfamily. Within this family of proteins exists a potent regulator known as myostatin (also called Growth Differentiation Factor 8, or GDF-8). Myostatin is secreted by skeletal muscle cells and acts autocrinely to inhibit muscle growth, ensuring that muscles do not grow too large for the skeletal and cardiovascular systems to support.
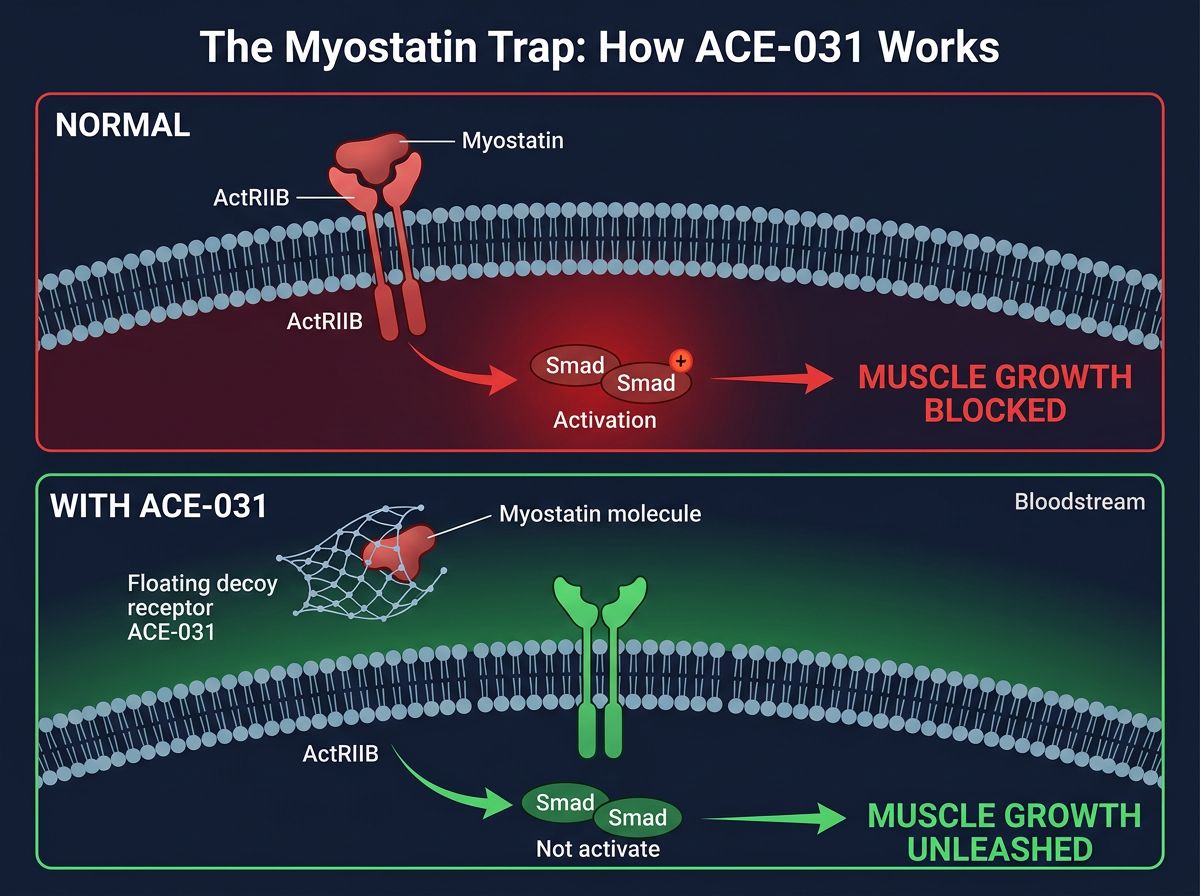
Under normal physiological conditions, myostatin binds to the Activin Receptor Type IIB (ActRIIB) on the surface of muscle cells. This binding initiates a signaling cascade involving Smad proteins, which ultimately translocate to the nucleus to downregulate the genes responsible for muscle protein synthesis and myogenesis. This is a negative feedback loop: as muscle mass increases, myostatin levels typically rise to cap further growth.
Genetic mutations that suppress myostatin—such as those seen in "mighty mice" or Belgian Blue cattle—result in a phenotype known as "double muscling," or extreme muscular hypertrophy. This suggests that the upper limit of muscle mass is not necessarily a metabolic ceiling, but a signaling ceiling imposed by myostatin. Breaking this ceiling has become a primary target for research into muscular dystrophy, sarcopenia, and cachexia.
ACE-031: The Soluble Decoy Receptor
Mechanism of Action
ACE-031 is a recombinant fusion protein. Structurally, it consists of the extracellular portion of the human Activin Receptor Type IIB (ActRIIB) fused to the Fc portion of human Immunoglobulin G1 (IgG1). This design is highly specific: the ActRIIB portion mimics the receptor found on muscle cells, while the Fc portion provides stability and prolongs the half-life of the molecule in the bloodstream.
The mechanism is elegant in its simplicity. When introduced into a biological system, ACE-031 circulates freely in the plasma. Myostatin and other related ligands (such as GDF-11 and Activin A) bind to the soluble ACE-031 instead of binding to the actual ActRIIB receptors on the muscle cell surface. ACE-031 acts as a "ligand trap" or a decoy. By sequestering these negative regulators in the bloodstream, ACE-031 prevents the activation of the downstream Smad signaling pathway.
Beyond Myostatin: Broad-Spectrum Inhibition
What distinguishes ACE-031 from a simple anti-myostatin antibody is its promiscuity regarding ligands. The ActRIIB receptor acts as a gateway for several members of the TGF-β superfamily, not just myostatin. Activin A and Activin B, for instance, also act effectively as negative regulators of muscle mass. An antibody targeting only myostatin might leave Activin A free to bind to the receptor, potentially blunting the hypertrophic effect.
ACE-031 blocks myostatin, GDF-11, and the Activins simultaneously. Research suggests that this broad-spectrum inhibition leads to substantially greater increases in muscle mass compared to myostatin inhibition alone. In animal studies, the administration of soluble ActRIIB-Fc has resulted in dramatic increases in lean body mass—up to 60% in some rodent models—within a matter of weeks.
Research Findings and Clinical Observations
Clinical trials involving ACE-031, particularly those focused on Duchenne Muscular Dystrophy (DMD), have demonstrated its potency. In a study by Attie et al. (2013), healthy postmenopausal women administered ACE-031 showed significant increases in lean muscle mass and thigh muscle volume after a single dose. The effects were rapid, occurring alongside a decrease in serum biomarkers of bone resorption and an increase in bone formation markers, suggesting a dual anabolic effect on muscle and bone.
However, the research landscape also highlights safety signals. The "decoy" nature of ACE-031 means it may trap ligands involved in other physiological processes. For example, some human trials were halted due to adverse events related to nosebleeds and gum bleeding (telangiectasia). This is hypothesized to be interference with ALK1 signaling, another pathway involved in vascular structure, which shares ligands with the ActRIIB pathway. Consequently, current research is often directed toward refining the specificity of the molecule or adjusting dosing protocols to mitigate off-target vascular effects.
TB-500: The Structural Engineer
Mechanism of Action: Actin Sequestration
While ACE-031 releases the brakes on size, TB-500 focuses on the internal scaffolding of the cell. TB-500 is a synthetic peptide fragment (amino acids 17-23) of the naturally occurring protein Thymosin Beta-4 (Tβ4). Tβ4 is ubiquitous in human cells and is particularly concentrated in wound fluid and platelets.
The primary mechanism of TB-500 is its interaction with actin, a protein that forms the microfilaments of the cytoskeleton. Actin exists in two forms: Globular actin (G-actin), which is free-floating monomeric sub-units, and Filamentous actin (F-actin), which forms the structural strands.
TB-500 functions as an actin-sequestering molecule. It binds to G-actin, maintaining a pool of ready-to-use monomers. When a cell needs to migrate, divide, or repair its structure, TB-500 releases these monomers to allow for rapid polymerization into F-actin. This process is critical for cell motility—the ability of cells to move to a site of injury—and structural remodeling.
Angiogenesis and Cell Migration
Beyond actin dynamics, research indicates that TB-500 significantly influences cell migration and angiogenesis (the formation of new blood vessels). Studies have shown that Tβ4 (and by extension, TB-500) can upregulate the expression of Vascular Endothelial Growth Factor (VEGF). This is vital for muscle tissue, specifically during hypertrophy.
As muscle fibers increase in size (hypertrophy), the diffusion distance for oxygen and nutrients from existing capillaries increases. Without a concurrent increase in capillary density (angiogenesis), the enlarged muscle tissue risks becoming hypoxic. TB-500's ability to promote endothelial cell migration and tube formation makes it a subject of interest for supporting the vascular needs of growing tissue.
Modulation of Inflammation
Another distinct property of TB-500 is its anti-inflammatory capability. It has been shown to downregulate inflammatory cytokines and prevent excessive fibrosis (scar tissue formation). In the context of muscle injury or extreme training stress, fibrosis can limit the contractility and flexibility of the muscle. By promoting organized tissue regeneration rather than disorganized scarring, TB-500 aids in maintaining the functional quality of the muscle tissue.
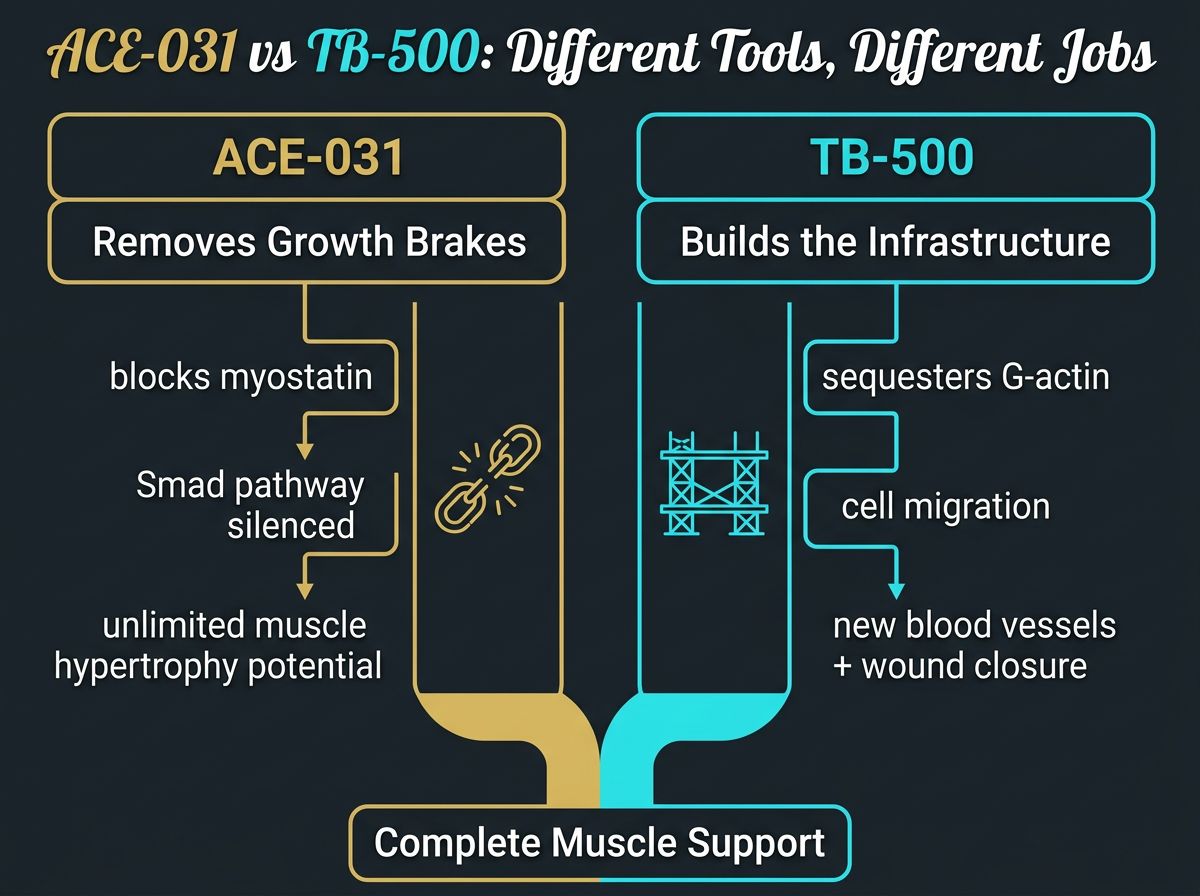
Comparative Analysis: The "Brake" vs. The "Builder"
To differentiate these two peptides clearly, it is helpful to use an analogy of construction. If building muscle is like constructing a skyscraper:
- ACE-031 removes the zoning laws that limit the building's height. It allows the structure to grow upward rapidly without regulatory restriction.
- TB-500 supplies the steel beams (actin) and the logistical routes (blood vessels) to ensure the building doesn't collapse under its own weight or suffer from lack of supplies.
Table 1: Comparative Profile of ACE-031 and TB-500
| Feature | ACE-031 (ActRIIB-Fc) | TB-500 (Thymosin Beta-4 Fragment) |
|---|---|---|
| Primary Class | Myostatin/Activin Inhibitor (Decoy Receptor) | Regenerative Peptide (Actin-Sequestering) |
| Mechanism | Ligand trapping (Myostatin, GDF-11, Activin A) | Upregulation of actin, promotion of cell migration |
| Primary Outcome | Rapid Skeletal Muscle Hypertrophy | Tissue Repair, Angiogenesis, Cytoskeletal Maintenance |
| Target Receptor | Soluble decoy for Activin Type IIB | Binds directly to G-Actin; interacts with ATP synthase |
| Effect on Strength | Increases absolute strength via mass accretion | Restores function; may improve endurance/flexibility |
| Vascular Effect | Anti-angiogenic (potential side effect via ALK1) | Pro-angiogenic (promotes VEGF expression) |
The "Uncoupling" of Mass and Function
A critical nuance in research involving ACE-031 and other myostatin inhibitors is the concept of "specific force." While ACE-031 generates massive muscles, some studies suggest that the generated muscle fibers may not be as densely packed with contractile proteins (myosin/actin) relative to their size as naturally trained muscle, or they may lack the mitochondrial density required for endurance. The muscle grows faster than the supporting infrastructure can adapt.
This is where the contrast with TB-500 is most stark. TB-500 does not induce hypertrophy directly. However, by supporting actin polymerization and vascular growth, it addresses the very deficits that rapid hypertrophy might create. It supports the "quality" of the tissue rather than the "quantity."
Synergistic Potential in Research Models
Given their opposing but potentially complementary mechanisms, theoretical research models often explore the intersection of these two pathways. The hypothesis is that combining a myostatin inhibitor (to drive size) with a regenerative agent (to support structure) could produce superior outcomes compared to either agent alone.
Addressing the Angiogenic Gap
One of the limitations of ACE-031 observed in early clinical trials was minor mucosal bleeding, theorized to be caused by cross-reactivity with BMP9/10 ligands that signal through ALK1—a pathway critical for keeping blood vessels stable. If ACE-031 accidentally "decoy-traps" ligands necessary for vascular health, it could lead to fragility in the capillaries.
TB-500 is inherently pro-angiogenic. In a hypothetical research setting, the presence of TB-500 might counteract the vascular instability potentially caused by broad-spectrum TGF-beta inhibition. By actively promoting endothelial cell migration and vessel stabilization, TB-500 could provide a protective effect against the vascular side effects of ACE-031. Note that this is currently a theoretical application based on mechanistic data and has not been fully validated in large-scale clinical trials.
Connective Tissue Integration
Rapid muscle growth places immense strain on tendons and ligaments. The muscle belly may strengthen faster than the connective tissue can adapt, leading to ruptures or tendonitis. TB-500 is well-documented for its role in soft tissue repair, particularly in tendons and ligaments. Co-administration in animal models suggests that while ACE-031 accelerates the contractile tissue growth, agents like TB-500 (or BPC-157) assist in the concomitant strengthening of the connective tissue matrix, preventing injury during the rapid growth phase.
Scientific Rigor: Methods and Quality Control
In executing studies involving these peptides, the purity and stability of the compounds are paramount. Both ACE-031 and TB-500 intricate molecules that are sensitive to degradation.
Administration Protocols in Literature
In peer-reviewed literature, dosing protocols vary significantly between rodent and primate models. ACE-031 acts systemically and has a longer half-life due to its Fc-fusion structure (often several days to a week in human subjects). Conversely, TB-500 has a very short plasma half-life but a persistent biological effect due to its cellular uptake and influence on gene expression.
- ACE-031: Typically administered subcutaneously. Research doses in murine models often range from 1mg/kg to 10mg/kg, administered once or twice weekly.
- TB-500: Administered subcutaneously or intraperitoneally in older studies. Dosing relies on "loading phases" in many anecdotal protocols, but clinical research usually utilizes steady, lower daily or every-other-day dosing to maintain elevated systemic levels of Tβ4.
Purity and Synthesis
For outcomes to be reproducible, researchers must utilize peptides synthesized with high fidelity. For ACE-031, the glycosylation of the protein (the attachment of carbohydrate groups) is critical for its stability and binding affinity. Improperly synthesized ACE-031 may degrade too quickly or fail to bind myostatin effectively. For TB-500, the specific sequence must be preserved to ensure interaction with G-actin.
Researchers should always verify their materials through rigorous testing. At Alpha Carbon Labs, we prioritize transparency in peptide synthesis. We encourage researchers to review our COA Documents to verify the purity and mass spectral analysis of the peptides used in their experiments. Ensuring the absence of trifluoroacetic acid (TFA) salts and endotoxins is also critical for in vivo studies to prevent confounding inflammatory responses.
Safety Considerations and Future Directions
While the potential for muscular dystrophy treatment and sarcopenia reversal is immense, safety remains the primary hurdle for clinical approval. The TGF-beta superfamily is involved in a vast array of physiological processes, including tumor suppression and reproductive health. Long-term inhibition of ActRIIB could theoretically impact FSH levels (affecting fertility) or allow premalignant cells to proliferate unchecked, although this has not been definitively observed in short-term studies.
For TB-500, the safety profile appears more benign in animal models, likely because it is a fragment of a protein already abundant in the body. However, because it promotes angiogenesis and cell migration, there is a theoretical concern regarding its use in subjects with active malignancies, as it could potentially facilitate tumor metastasis (though Tβ4 itself acts differently in different tissue contexts).
The future of this research likely lies in specificity. The next generation of myostatin inhibitors aims to bind only myostatin and GDF-11, sparing the Activins and BMPs to avoid vascular and reproductive side effects. Meanwhile, combination therapies utilizing repair agents like TB-500 alongside growth agents are likely to become a standard approach in addressing complex musculoskeletal disorders.
Conclusion
The comparison between ACE-031 and TB-500 is not a matter of which is "better," but rather which physiological lever is being pulled. ACE-031 represents the cutting edge of genetic modulation—a tool to bypass the natural homeostatic limits of muscle mass by silencing the myostatin signaling pathway. It is a peptide of profound power and specific utility in hypertrophy research.
TB-500 represents the foundational support—an agent of repair, maintenance, and structural integrity. It ensures that the cellular environment is conducive to survival, movement, and adaptation. In the quest to treat muscle wasting diseases or enhance physical capacity, these two peptides illustrate the necessary balance between the impulse to grow and the necessity to sustain that growth.
For researchers aiming to push the boundaries of musculoskeletal science, understanding the interplay between the Activin receptor pathway and cytoskeletal actin dynamics provides a roadmap for developing more effective, holistic therapies for muscle pathologies.
References
- 1. Attie, K. M., et al. "A single ascending-dose study of muscle-regulating effects of ACE-031 in healthy volunteers." Muscle & Nerve, vol. 47, no. 3, 2013, pp. 416-423.
- 2. Philp, D., et al. "Thymosin beta4 and its degradative product, Ac-SDKP, are novel angiogenic agents." Blood, vol. 101, no. 12, 2003, pp. 4867-4874.
- 3. Lee, S. J., & McPherron, A. C. "Regulation of myostatin activity and muscle growth." Proceedings of the National Academy of Sciences, vol. 98, no. 16, 2001, pp. 9306-9311.
- 4. Goldstein, A. L., et al. "Thymosin β4: actin-sequestering protein moonlights to repair injured tissues." Trends in Molecular Medicine, vol. 18, no. 1, 2012, pp. 238-253.
- 5. Campbell, C., et al. "Myostatin inhibition using ACE-031 in Duchenne muscular dystrophy: A randomized placebo-controlled trial." Muscle & Nerve, vol. 55, no. 4, 2017, pp. 458-464.
- 6. Huff, T., et al. "Thymosin beta-4 release from human platelets." Journal of Pharmaceutical and Biomedical Analysis, vol. 53, no. 3, 2010, pp. 325-334.
- 7. Trendelenburg, A. U., et al. "Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size." American Journal of Physiology-Cell Physiology, vol. 296, no. 5, 2009, pp. C1258-C1270.
- 8. Smart, N., et al. "Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization." Nature, vol. 445, no. 7124, 2007, pp. 177-182.
- 9. Tsuchida, K. "Targeting myostatin for muscle growth and neuromuscular disorders." Current Opinion in Pharmacology, vol. 34, 2017, pp. 9-14.
- 10. Klein, J. D., et al. "Thymosin beta 4 improves cardiac function after myocardial infarction in mice." Journal of Molecular and Cellular Cardiology, vol. 39, no. 5, 2005, pp. 863-871.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.