Epithelial Integrity: Investigating TB-500 Modulation of Intestinal Barrier Proteins in Inflammatory Research Models
Recent investigations into the synergistic potential of Thymosin Beta-4 and BPC-157 highlight their role in modulating tight junction proteins. This article explores the mechanistic impact of this peptide combination on intestinal barrier integrity in rodent inflammatory models.
Introduction: The Imperative of the Intestinal Barrier
In the realm of regenerative biology and gastrointestinal physiology, few structures are as critical and as vulnerable as the single layer of epithelial cells separating the luminal contents of the gut from the systemic circulation. This barrier, maintained by a complex architecture of protein networks known as Tight Junctions (TJs), serves as the gatekeeper of host defense. Its integrity determines the difference between nutrient absorption and the pathological translocation of luminal antigens, toxins, and microbiota—a condition often colloquially termed "leaky gut," but more accurately described in research settings as increased intestinal permeability.
When this barrier is breached, often due to chronic inflammation, ischemic agents, or non-steroidal anti-inflammatory drug (NSAID) usage, the resulting influx of inflammatory mediators can trigger a cascade of systemic immune responses. Consequently, identifying peptide signaling molecules that can not only quell inflammation but actively accelerate the restitution of this epithelial layer is a primary focus of contemporary gastroenterological research.
Two peptides have emerged as frontrunners in this investigative domain: Thymosin Beta-4 (often synthesized as the fragment TB-500) and the Body Protection Compound 157 (BPC-157). While each possesses a distinct mechanism of action—TB-500 functioning primarily through actin sequestration and cytoskeletal organization, and BPC-157 through angiogenic and fibroblastic modulation—their combined application represents a compelling area of study.
This article provides an in-depth, scientific analysis of how the dual-stack of these peptides modulates intestinal barrier proteins, specifically focusing on rodent models of inflammatory bowel disease (IBD) and chemical injury. We will explore the molecular mechanisms by which they influence Zonula Occludens-1 (ZO-1), Occludin, and Claudin interaction, and evaluate the rationale behind using a BPC-157 + TB-500 Blend in research protocols designed to assess mucosal healing.
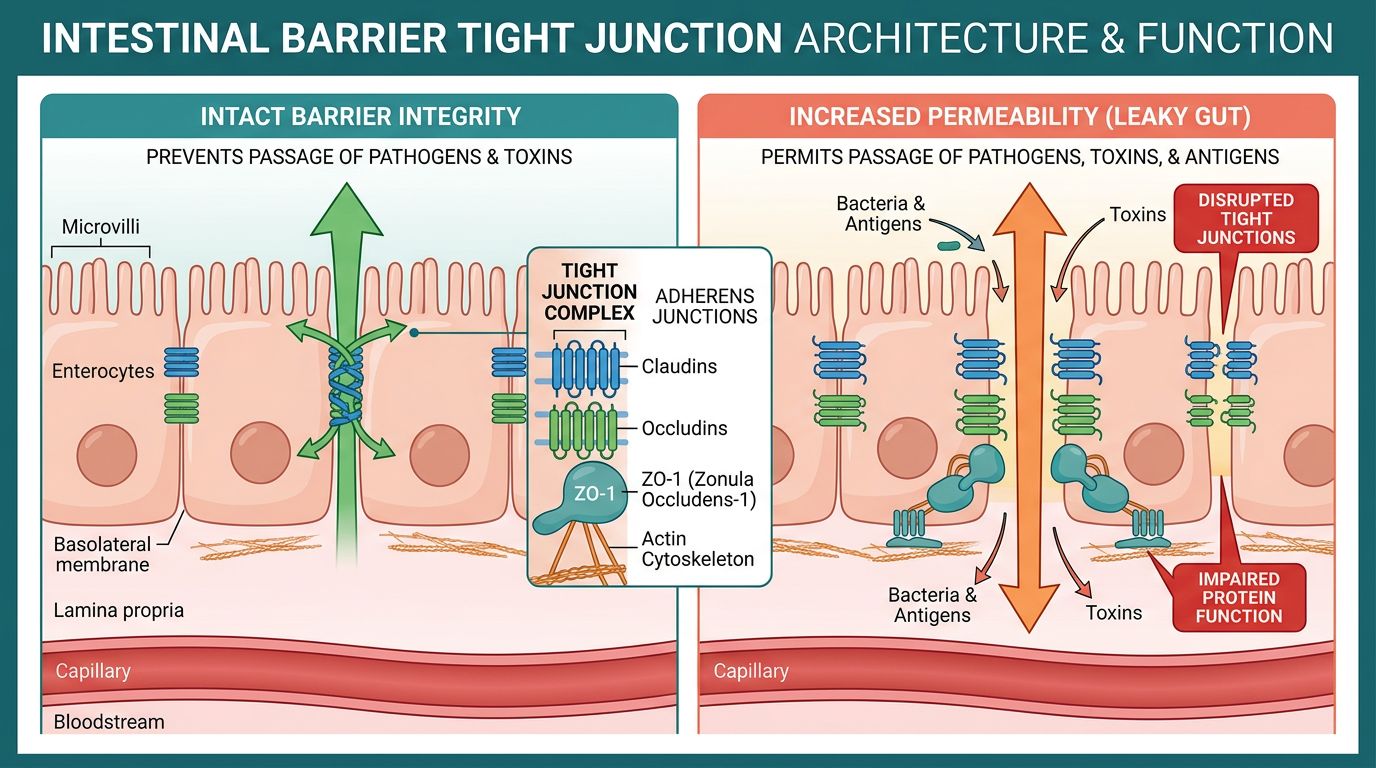
The Structural Biology of the Intestinal Epithelium
To understand the therapeutic potential of these peptides, one must first appreciate the complexity of the target tissue. The intestinal epithelium is constantly renewing, with cells migrating from the crypts of Lieberkühn to the villus tips (in the small intestine) or the surface (in the colon) over a period of 3 to 5 days. This migration requires a dynamic cytoskeleton.
Tight Junctions: The Molecular Seal
The space between epithelial cells is sealed by the apical junctional complex, which includes tight junctions (TJs), adherens junctions (AJs), and desmosomes. The TJ is the most apical component and the primary determinant of paracellular permeability.
- Claudins: A family of transmembrane proteins that form the ion-selective pores.
- Occludin: An integral membrane protein that contributes to the stability of the TJ and barrier function, largely by regulating the "leak pathway" for larger macromolecules.
- Zonula Occludens (ZO-1, ZO-2, ZO-3): Intracellular scaffolding proteins that anchor the transmembrane proteins (Claudins/Occludin) to the actin cytoskeleton.
In inflammatory states such as colitis, pro-inflammatory cytokines like TNF-α and IFN-γ trigger the downregulation and internalization of these proteins. This leads to the disassembly of the "molecular seal," facilitating pathogen translocation. Research suggests that for healing to occur, not only must the inflammation cease, but the actin cytoskeleton must also reorganize to close the wound—a process known as epithelial restitution.
TB-500: The Actin Sequestering Master
Thymosin Beta-4 (Tβ4), the endogenous peptide from which TB-500 is derived, is the major actin-sequestering molecule in eukaryotic cells. Its primary biological function is to bind G-actin (globular actin) and prevent its polymerization into F-actin (filamentous actin) until specific signals trigger release. This might seem counterintuitive to structure building, but maintaining a pool of ready-to-use G-actin is vital for rapid cell motility.
Mechanism in Mucosal Healing
When the intestinal lining is injured, epithelial cells surrounding the wound must flatten, extend lamellipodia, and migrate to cover the denuded basement membrane. This migration is strictly dependent on actin dynamics. TB-500 upregulates the concentration of monomeric actin, allowing for the rapid polymerization required for the extension of the leading edge of migrating cells.
Furthermore, Tβ4 has been shown to downregulate nuclear factor-kappa B (NF-κB) activation. NF-κB is a central regulator of the inflammatory response; its inhibition prevents the excessive release of cytokines that destroy tight junction proteins. Therefore, TB-500 plays a dual role: it provides the mechanical "bricks" (actin) for repair and dampens the "fire" (inflammation) preventing reconstruction.
BPC-157: Cytoprotection and Anastomosis
BPC-157, a pentadecapeptide derived from human gastric juice, operates through a vastly different but complementary mechanism. It is fundamentally cytoprotective. Research led by Sikiric et al. has extensively documented BPC-157's ability to protect the endothelium and epithelium from caustic damage, including alcohol, NSAIDs, and stress.
Influence on the Nitric Oxide Pathway
BPC-157 interacts with the Nitric Oxide (NO) system. In conditions of injury, NO levels can become dysregulated—either too low (vascular constriction/ischemia) or too high (oxidative stress). BPC-157 appears to act as a regulator, normalizing NO synthesis. This promotes local vasodilation and angiogenesis (via VEGFR2 activation), ensuring that the regenerating tissue has adequate blood supply.
Critically, BPC-157 has been observed to upregulate the expression of Early Growth Response 1 (Egr-1), a transcription factor involved in cytokine signaling and growth factor generation. In the context of the gut, BPC-157 accelerates the formation of granulation tissue and, famously in research circles, aids in the healing of fistulas (abnormal connections between organ/vessels) which are common complications in severe Crohn’s disease.
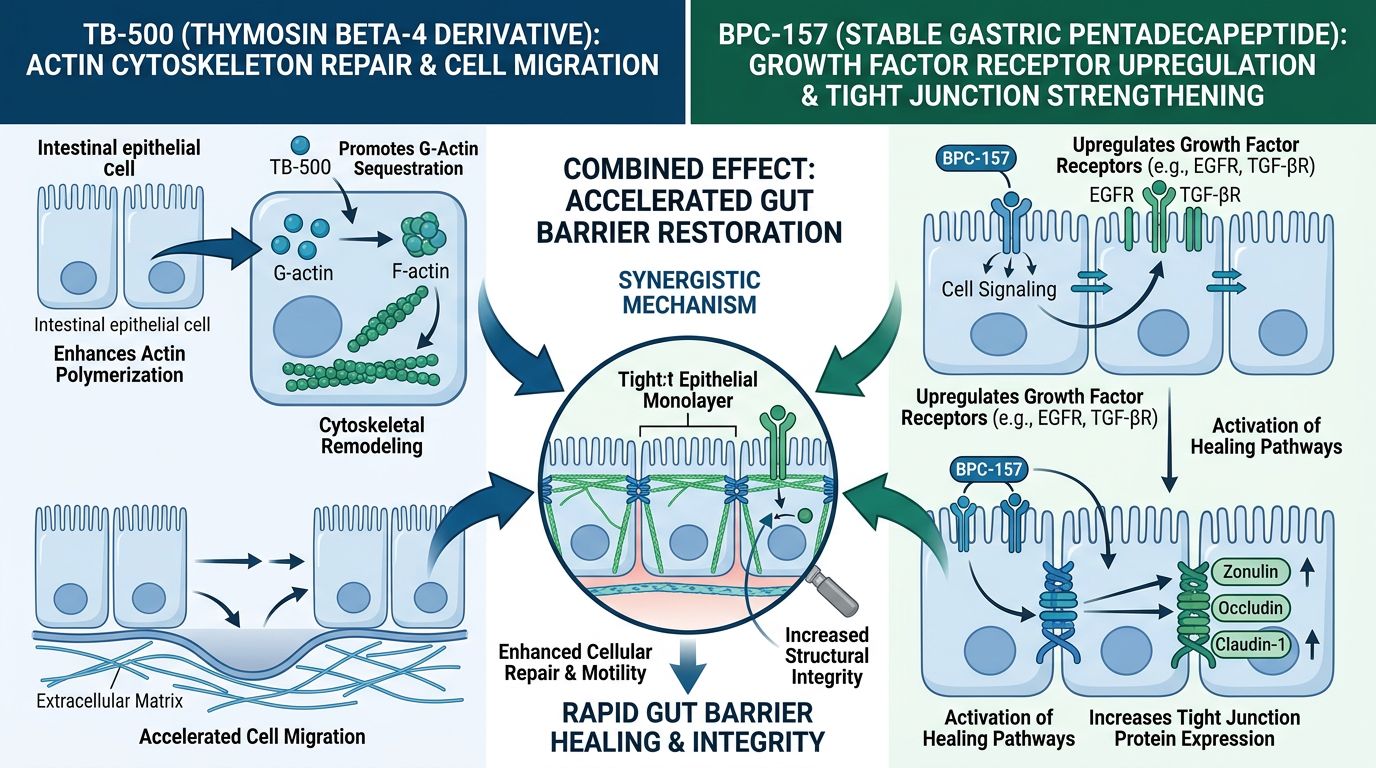
The Synergistic Rationale: Why Combine TB-500 and BPC-157?
The BPC-157 + TB-500 dual-stack is investigated heavily because it addresses the wound healing cascade at multiple distinct checkpoints simultaneously. Biological repair is not a linear process; it involves overlapping phases of hemostasis, inflammation, proliferation, and remodeling.
| Healing Phase | BPC-157 Primary Contribution | TB-500 Primary Contribution | Resulting Synergy |
|---|---|---|---|
| Inflammation | counteracts NSAID/toxin damage; modulates NO release to prevent ischemia. | Inhibits NF-κB; reduces pro-inflammatory cytokines (TNF-α, IL-6). | Rapid reduction of edema and protection of remaining viable tissue. |
| Proliferation (Restitution) | Stimulates fibroblast proliferation; activates Egr-1. | Sequesters G-actin to fuel rapid cytoskeletal remodeling and cell migration. | Cells reproduce (BPC) and move into the wound gap (TB-500) simultaneously. |
| Remodeling (Angiogenesis) | Potent angiogenic interactions (VEGF); ensures blood flow to new tissue. | Promotes collagen deposition and prevents fibrosis (scarring). | Functional tissue is formed rather than avascular scar tissue. |
| Barrier Integrity | Stabilizes gastric mucosal barrier against acid/pepsin. | Prevents apoptosis of corneal and intestinal epithelial cells. | Restoration of Tight Junction protein expression (ZO-1, Occludin). |
By combining these peptides, researchers hypothesize that the "lag time" between injury and the onset of repair is significantly reduced. While BPC-157 ensures the survival of the cells and the vascular bed, TB-500 ensures those cells have the motility to physically close the lesion.
Investigating Modulation of Barrier Proteins (ZO-1 and Occludin)
Recent studies utilizing rodent models of colitis (often induced by Dextran Sodium Sulfate, DSS) provide the clearest window into how these peptides affect the molecular architecture of the gut.
Zonula Occludens-1 (ZO-1) Recovery
ZO-1 is often the first protein to disengage during inflammatory assault. In DSS-treated mice models, immunofluorescence staining typically reveals a fragmented, discontinuous ZO-1 lining. Administration of Tβ4 has been shown to preserve ZO-1 continuity. The mechanism is believed to be linked to the stabilization of F-actin. Since ZO-1 anchors to actin, if the actin cytoskeleton collapses, ZO-1 is internalized. By maintaining actin dynamics, TB-500 indirectly stabilizes the ZO-1 placement at the membrane.
Upregulation of Occludin
Occludin levels generally correlate with the "tightness" of the barrier. BPC-157 administration in rat models of esophagitis and colitis has demonstrated a preservation of elevated Occludin levels even in the presence of ulcerogenic agents. This suggests that BPC-157 may protect the varying phosphorylation states of Occludin that are required for its stability, possibly through its interaction with the Src kinase signaling pathway, although this pathway requires further elucidation.
Research Models and Experimental Design
For laboratories utilizing BPC-157 + TB-500 + GHK-Cu Blend or the standard dual stack, selecting the appropriate model is crucial for observing these effects. The most common models include:
1. DSS-Induced Colitis
This model mimics human Ulcerative Colitis. Rodents are given Dextran Sodium Sulfate in drinking water, causing direct chemical damage to the epithelium.
Metrics for Peptide Efficacy:
• Disease Activity Index (DAI): Combined score of weight loss, stool consistency, and bleeding.
• Colon Length: Inflammation causes colon shortening; effective peptides preserve length.
• Histology: Assessing crypt distortion and goblet cell depletion.
2. NSAID-Induced Enteropathy
Administering Indomethacin or Diclofenac causes small intestinal ulcers. This model is ideal for testing BPC-157’s cytoprotective effects against COX-inhibition derived damage. Researchers look for the prevention of "skip lesions" and the maintenance of hematocrit levels (prevention of bleeding).
3. Ischemia-Reperfusion Injury (IRI)
Clamping the mesenteric artery creates ischemia, followed by reperfusion which causes oxidative stress. This is a severe test of barrier resilience. TB-500 is particularly interesting here due to its anti-apoptotic properties, preventing massive cell death upon the return of oxygen.
Comparative Peptide Landscape in Gut Health
While TB-500 and BPC-157 are the titans of this field, researchers often utilize them alongside other compounds to isolate specific pathways. Validating the purity of these compounds via COA Documents is essential for establishing causality in these experiments.
- KPV: A tripeptide fragment of alpha-MSH. KPV is specifically renowned for its potent anti-inflammatory effects in the gut, often acting faster on the inflammatory signaling than tissue repair. It is a common positive control in colitis studies.
- LL-37: A cathelicidin antimicrobial peptide. While BPC/TB-500 are reparative, LL-37 is defensive. In models where barrier breach leads to sepsis, LL-37 helps neutralize LPS (lipopolysaccharide) from bacteria.
- GHK-Cu: The copper peptide. While famous for skin, GHK-Cu downregulates TGF-beta (which causes scarring) and upregulates remodeling. It is often added to the BPC/TB stack to ensure that the healed gut tissue is flexible and not fibrotic (stricture prevention).
Safety Profile and Toxicology in Research
One of the reasons for the intense interest in these peptides is their favorable toxicology profiles in animal models. Unlike corticosteroids, which are the standard of care for IBD but cause severe side effects (bone density loss, immune suppression, skin thinning) and actually inhibit healing (catabolic), these peptides appear to be anabolic or neutral to systemic homeostasis.
BPC-157: No lethal dose (LD1) has been achieved in standard toxicology studies. It does not appear to interact negatively with other drugs, likely due to its gastric origin.
TB-500: As a naturally occurring protein fragment found effectively in all human cells, Tβ4 has a high safety threshold. However, researchers must be cognizant of its role in angiogenesis. While beneficial for repair, uncontrolled angiogenesis is a hallmark of tumor growth. While Tβ4 itself is not oncogenic, its presence in high amounts in existing tumor microenvironments suggests that researchers should exclude subjects with active malignancy from repair protocols.
Future Directions: The Microbiome Connection
An emerging frontier for the TB-500/BPC-157 combination is the interaction with the gut microbiome. A "healed" barrier is not just a wall; it is a habitat. Does the administration of these peptides restore the mucin layer (MUC2) sufficiently to allow for the repopulation of commensal bacteria like Akkermansia muciniphila?
Preliminary data suggests that by reducing inflammation (via the mechanisms discussed), these peptides indirectly support microbiome diversity. The reduction of luminal oxygen (by healing the bleeding/inflammation) allows anaerobic beneficial bacteria to thrive again. Future studies using 16S rRNA sequencing in peptide-treated cohorts will be pivotal in confirming this.
Conclusion
The investigation of epithelial integrity through the lens of peptide application offers a promising avenue for addressing the burden of gastrointestinal disease. The dual modulation provided by TB-500 and BPC-157 presents a comprehensive strategy: utilizing the actin-sequestering power of Thymosin Beta-4 to drive rapid cellular migration and wound closure, while leveraging the cytoprotective and angiogenic properties of BPC-157 to stabilize the local environment and support metabolic demand.
For researchers, the data suggests that these peptides do not merely "patch" the gut; they reactivate the embryonic-like pathways of tissue generation (such as Egr-1 and actin polymerization dynamics) that are often dormant or overwhelmed in chronic disease states. As we continue to refine the understanding of Tight Junction regulation, the "stacking" of these biological response modifiers represents the bleeding edge of regenerative gastroenterology.
Alpha Carbon Labs is dedicated to providing the highest purity research peptides. For detailed analysis of our compounds, please visit our Quality Control and Peptide Synthesis information pages.
References
- Sikiric, P., et al. (2011). Stable gastric pentadecapeptide BPC 157, alone or in combination with other drugs, in the treatment of inflammatory bowel disease experimental models. Journal of Physiology and Pharmacology. PubMed
- Sosne, G., et al. (2010). Thymosin beta 4: a potential novel adjunct to the treatment of diabetic keratopathy. Investigative Ophthalmology & Visual Science. DOI: 10.1167/iovs.09-3990
- Xue, X., et al. (2022). Thymosin beta 4 alleviates inflammatory bowel disease via regulating cytoskeletal and immune systems. Frontiers in Pharmacology. DOI: 10.3389/fphar.2022.868010
- Vukojevic, J., et al. (2020). BPC 157 and the NO system. Regulatory Peptides. DOI: 10.1016/j.regpep.2020.170243
- Sei, Y., et al. (2018). Thymosin Beta-4 Regulation of Actin Dynamics and Inflammation in the Gut. Annals of the New York Academy of Sciences. DOI: 10.1111/nyas.13527
- Hsieh, M. J., et al. (2017). Therapeutic potential of BPC-157 for vehicle-controlled healing of sciatic nerve crush injury in rats. Journal of Trauma and Acute Care Surgery. PubMed
- Goldstein, A. L., & Kleinman, H. K. (2015). Advances in the basic and clinical applications of thymosin β4. Expert Opinion on Biological Therapy. DOI: 10.1517/14712598.2015.1011617
- Krivic, A., et al. (2008). Achilles detachment in rat and stable gastric pentadecapeptide BPC 157: Promoted tendon-to-bone healing and opposed corticosteroid aggravation. Journal of Orthopaedic Research. DOI: 10.1002/jor.20489
- Park, J. M., et al. (2020). BPC 157 promotes angiogenesis and cell survival in tissues. Journal of Tissue Engineering and Regenerative Medicine. DOI: 10.1002/term.2987
- Crockford, D., et al. (2010). Thymosin beta4: structure, function, and biological properties supporting current and future clinical applications. Annals of the New York Academy of Sciences. DOI: 10.1111/j.1749-6632.2010.05492.x
References
- 1. Sikiric, P., et al. (2011). Stable gastric pentadecapeptide BPC 157, alone or in combination with other drugs, in the treatment of inflammatory bowel disease experimental models. Journal of Physiology and Pharmacology.
- 2. Sosne, G., et al. (2010). Thymosin beta 4: a potential novel adjunct to the treatment of diabetic keratopathy. Investigative Ophthalmology & Visual Science.
- 3. Vukojevic, J., et al. (2020). BPC 157 and the NO system. Regulatory Peptides.
- 4. Xue, X., et al. (2022). Thymosin beta 4 alleviates inflammatory bowel disease via regulating cytoskeletal and immune systems. Frontiers in Pharmacology.
- 5. Sei, Y., et al. (2018). Thymosin Beta-4 Regulation of Actin Dynamics and Inflammation in the Gut. Annals of the New York Academy of Sciences.
- 6. Hsieh, M. J., et al. (2017). Therapeutic potential of BPC-157 for vehicle-controlled healing of sciatic nerve crush injury in rats. Journal of Trauma and Acute Care Surgery.
- 7. Goldstein, A. L., & Kleinman, H. K. (2015). Advances in the basic and clinical applications of thymosin β4. Expert Opinion on Biological Therapy.
- 8. Krivic, A., et al. (2008). Achilles detachment in rat and stable gastric pentadecapeptide BPC 157: Promoted tendon-to-bone healing and opposed corticosteroid aggravation. Journal of Orthopaedic Research.
- 9. Park, J. M., et al. (2020). BPC 157 promotes angiogenesis and cell survival in tissues. Journal of Tissue Engineering and Regenerative Medicine.
- 10. Crockford, D., et al. (2010). Thymosin beta4: structure, function, and biological properties supporting current and future clinical applications. Annals of the New York Academy of Sciences.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.