Beyond the Dual-Agonist Ceiling: Methodological Requirements for Transitioning Research Models to Retatrutide Triple-Agonism
Discover how transitioning from a dual-agonist to a triple-agonist like Retatrutide shatters weight loss plateaus. Learn the science behind upgrading your metabolic optimization protocols.
Beyond the Dual-Agonist Ceiling: Methodological Requirements for Transitioning Research Models to Retatrutide Triple-Agonism
For decades, the pursuit of optimal metabolic function and sustainable weight management has been the holy grail of peptide research. If you have been paying attention to the wellness, anti-aging, or fitness spaces over the last few years, you have undoubtedly witnessed the absolute explosion of metabolic peptides. Initially, it was all about controlling appetite and improving how the body manages blood sugar. But as our understanding of systemic biology has evolved, so too have the compounds we use to optimize it.
Today, researchers and everyday health-conscious individuals are encountering a fascinating phenomenon: the dual-agonist ceiling. While second-generation peptides have delivered phenomenal results, the human body is incredibly adaptive. Eventually, weight loss slows down. Energy levels plateau. The metabolic thermostat resets itself. This physiological resistance is precisely why the scientific community has developed a new class of metabolic optimization: the triple-agonist.
In this comprehensive guide, we are diving deep into what exactly happens when you move beyond a dual-hormone approach into the realm of triple-agonism. We will break down the molecular shifts, explain why the body plateaus on older peptides, and outline the specific requirements for successfully transitioning your research model (or your personal metabolic journey) from a dual-agonist to a triple-agonist powerhouse.
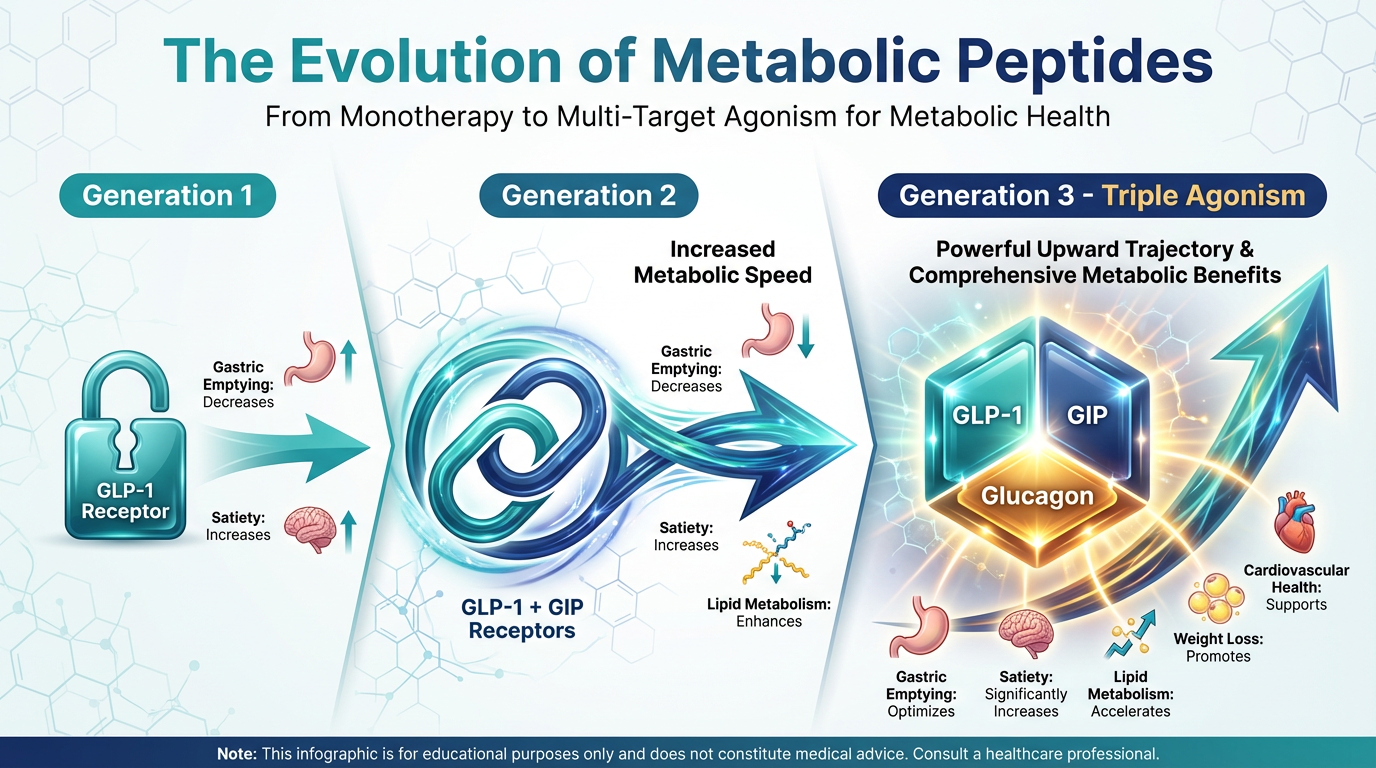
The Evolution of Metabolic Optimization: From Single to Triple Action
To truly appreciate how revolutionary triple-agonists are, we have to look at the stepping stones that got us here. The path of metabolic peptide development is a story of compounding synergy. Let's break down the “generations” of these peptides in plain English, so you can see exactly why the science had to keep advancing.
Generation 1: The Single Agonist (GLP-1)
The first massive breakthrough in this space came with single-agonist peptides, most notably Semaglutide. These peptides target one specific receptor in the body: the GLP-1 (Glucagon-Like Peptide-1) receptor.
In simple terms, GLP-1 does two main things for weight loss. First, it slows down gastric emptying, meaning food stays in your stomach longer and you feel full on significantly less food. Second, it sends a direct signal to the satiety centers of your brain, turning off the “food noise” and constant cravings. For millions of people, this single-pronged approach was enough to kickstart life-changing weight loss.
Generation 2: The Dual-Agonist (GLP-1 + GIP)
As incredible as single agonists are, researchers realized that combining GLP-1 with a second hormone could amplify the results while smoothing out some of the side effects. Enter Tirzepatide, the world’s first highly successful dual-agonist. Tirzepatide combines GLP-1 with GIP (Glucose-Dependent Insulinotropic Polypeptide).
Why was adding GIP such a big deal? While GLP-1 suppresses appetite, GIP actively works on insulin sensitivity and lipid (fat) metabolism. Even better, GIP naturally reduces the nausea often associated with strong GLP-1 medications. By hitting two receptors at once, Tirzepatide delivered much faster and more profound fat loss than its single-agonist predecessors. It was a massive leap forward in metabolic optimization.
Generation 3: The Triple-Agonist (GLP-1 + GIP + Glucagon)
This brings us to the bleeding edge of current metabolic science: Retatrutide. Retatrutide is a triple-agonist, affectionately known in the research community as a "tri-agonist." It takes the established magic of GLP-1 and GIP, and adds a third powerhouse hormone to the mix: Glucagon (GCG).
If you know a little bit about biology, the addition of glucagon might sound confusing. Normally, we think of glucagon as the hormone that raises blood sugar, which seems counterproductive for weight loss. However, when glucagon is combined synergistically with GLP-1 and GIP, it performs a metabolic miracle: it dramatically increases your basal metabolic rate (how many calories you burn just staying alive) and directly attacks visceral fat in the liver.
This molecular shift—pushing the body into an active fat-burning state rather than simply relying on starvation-level calorie restriction—is what shatters the ceiling of older peptides.
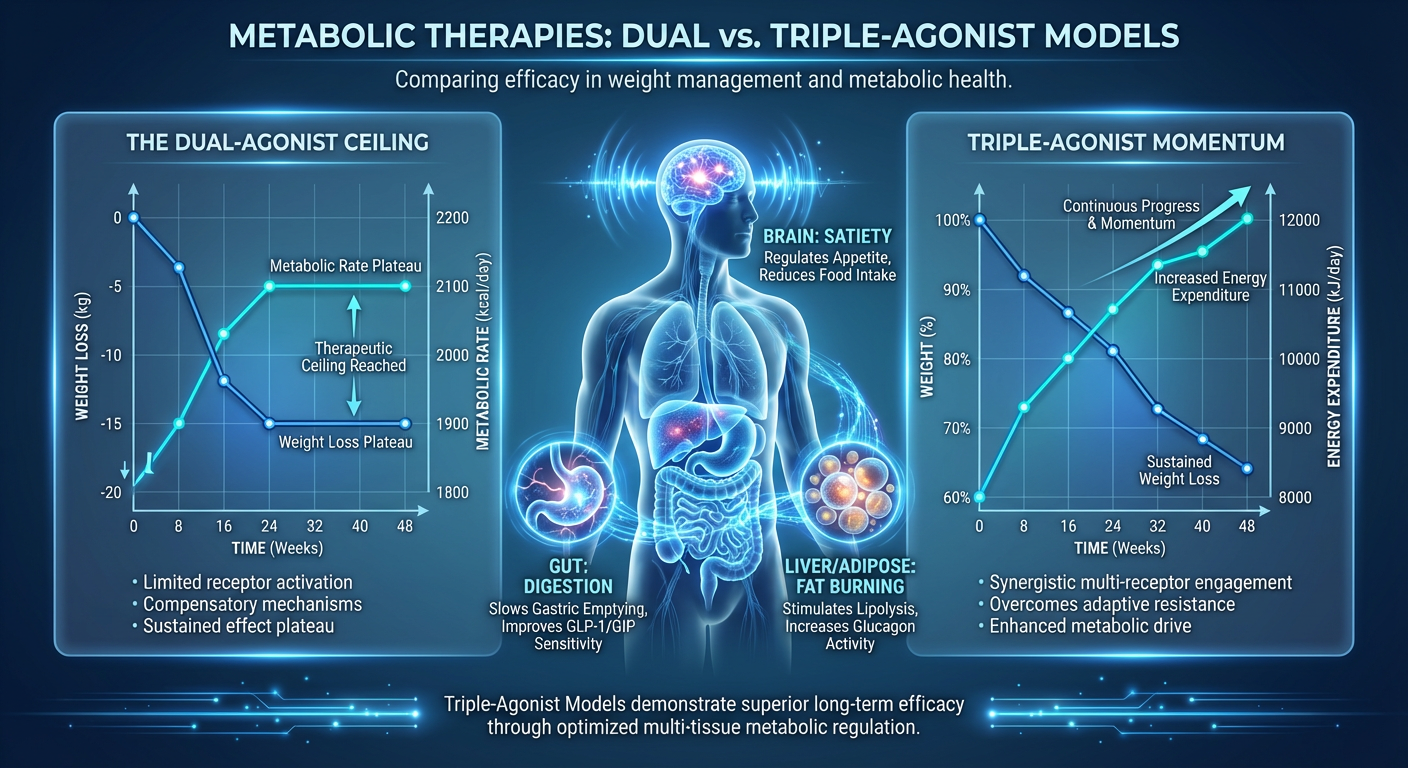
What is the Dual-Agonist Ceiling?
If dual-agonists are so effective, why do we need a triple-agonist? Why take the next step?
When you start researching or utilizing a dual-agonist protocol, the initial phase is often characterized by rapid, seemingly effortless fat loss. The appetite suppression kicks in abruptly, insulin sensitivity improves, and the scale drops predictably. However, human evolution is entirely based on survival, and the human body absolutely hates losing stored energy (fat).
Around the 8-to-12-month mark of a dual-agonist protocol, many individuals hit what is known as a plateau, or the "dual-agonist ceiling." Here is precisely what happens on a biological level:
- Metabolic Adaptation: As your body loses weight, your resting metabolic rate naturally plunges. A smaller body requires fewer calories to maintain itself. However, the body actually slows your metabolism down disproportionately to the amount of weight you’ve lost. It is trying to pump the brakes on fat loss to prevent starvation.
- Receptor Downregulation: Over an extended period of time, your GLP-1 and GIP receptors can become desensitized. The same dosage that once melted fat and crushed cravings suddenly feels like a sugar pill. The “food noise” creeps back in, and hunger returns.
- The Leptin Problem: Leptin is the hormone that tells your body you have enough fat stored. As you lose body fat, leptin levels drop. Low leptin tells your brain, “We are starving, stop burning energy and conserve fat.” Dual-agonists suppress appetite, but they do not directly alter the body's baseline energy expenditure enough to overcome a severe leptin drop.
In plain English, the dual-agonist ceiling is the point where eating less is no longer enough. Your metabolism has slowed down to match your low food intake. You are trapped in a standoff with your own biology. To break this ceiling, you do not need to eat even less—you need to signal the body to actively burn more energy.
The Molecular Shifts: What Happens When You Upgrade to Retatrutide?
When making the transition to a triple-agonist model, the biological environment undergoes a profound transformation. You are moving from a system of restriction (primarily eating less) to a system of active expenditure (burning more). Let's look at the three major molecular shifts that occur during this transition.
1. Unlocking the Glucagon Energy Furnace
The defining feature of a triple agonist is the activation of the glucagon receptor. While GLP-1 and GIP keep blood sugar stable and appetite suppressed, glucagon acts like a physiological thermostat turned all the way up. It stimulates energy expenditure. In clinical research phases, subjects utilizing triple agonists show a measurable increase in their basal metabolic rate. This means that even while sitting entirely still, the body is expending more calories than it was on a dual-agonist. This directly counteracts the metabolic slowdown that normally halts weight loss.
2. Aggressive Hepatic Fat Clearance
One of the most remarkable findings in phase 2 trials for triple agonists was their effect on the liver. Visceral fat—specifically liver fat (steatosis)—is incredibly dangerous and stubborn. It drives systemic inflammation, insulin resistance, and makes overall fat loss incredibly difficult.
Glucagon receptor agonism specifically targets liver fat. The triple-agonist molecular structure forces the liver to rapidly break down and utilize stored triglycerides. In research data, a vast majority of subjects using a triple-agonist saw their liver fat completely resolve in just a matter of months. A healthy liver processes circulating fats and sugars optimally, breaking the gridlock of metabolic syndrome.
3. Enhanced Lipid Metabolism
Beyond the liver, triple-agonists fundamentally alter lipid metabolism throughout the entire body. The combination of GIP and Glucagon forces white adipose tissue (stored belly or thigh fat) to release free fatty acids into the bloodstream to be burned as fuel. This means you aren't just losing water weight or muscle; the body is being chemically instructed to prefer stored body fat as its primary energy source.
Methodological Requirements for Transitioning Research Models
Transitioning from a well-established dual-agonist routine to a highly potent triple-agonist is not as simple as swapping one vial for another. Because the mechanism of action involves increasing energy expenditure rather than just suppressing appetite, the methodological requirements—or how you go about making this switch—must be managed carefully.
Whether you are a researcher designing a cellular model or a health enthusiast mapping out a longevity protocol, here are the step-by-step methodologies required for a safe, optimal transition.
Phase 1: Assessing Metabolic Readiness
Before introducing a triple agonist, the foundation must be secure. Have you genuinely hit the dual-agonist ceiling? If a research subject is still steadily losing body fat and tolerating a dual agonist well, there is no need to advance. The triple-agonist is the heavy artillery—reserve it for when the plateau has persisted for at least 4 to 6 weeks despite proper diet and exercise.
Phase 2: The Washout vs. Direct Titration Protocol
When moving from a peptide like Tirzepatide to Retatrutide, researchers typically look at two models:
- The Washout Method: Discontinuing the dual-agonist entirely, allowing the body to clear the peptide over 2-3 weeks, and then starting the triple-agonist at the absolute lowest baseline dose. This allows receptors a brief rest, though it may trigger a temporary rebound in hunger.
- The Cross-Titration Method: Lowering the dose of the existing dual-agonist while introducing a micro-dose of the triple-agonist, eventually fully replacing the former.
In most modern applications, a direct switch is done by calculating an equivalent starting dose. However, because Retatrutide involves the glucagon receptor, starting low and moving slow is non-negotiable. The increased metabolic rate can cause a harmless but noticeable increase in resting heart rate initially. Easing into the new peptide allows the cardiovascular system to acclimate comfortably.
Phase 3: Managing Energy Expenditure and Hydration
This is where the transition requires the most attention. Because your body is suddenly functioning exactly like a metabolic furnace, water and electrolyte utilization skyrocket. The glucagon receptor activation drastically increases lipid oxidation, a process that requires massive amounts of water.
A common mistake when moving to a triple-agonist is maintaining the same hydration protocols used on older peptides. Subjects can easily become dehydrated, which may present as fatigue, headaches, or muscle cramps. Increasing daily water intake by at least 30%, layered with high-quality sodium, potassium, and magnesium, is a strict methodological requirement for this protocol.
Phase 4: Protein Prioritization and Muscle Preservation
Because a triple-agonist drives up the body’s daily calorie burn, the risk of catabolism (burning muscle for energy) increases alongside fat burning. If you do not supply the body with adequate amino acids, it will break down lean tissue. Prioritizing incredibly high protein intakes is vital. In the anti-aging and optimization space, this is usually combined with heavy resistance training to send a strong "keep this muscle" signal to the brain.
Synergistic Peptides: Supporting the Triple-Agonist Framework
No peptide exists in a vacuum. As you transition to an advanced metabolic accelerator, utilizing supportive and synergistic peptides can mitigate side effects, protect muscle tissue, and enhance overall subjective well-being. Astute researchers often pair powerful fat-loss protocols with focused recovery compounds.
Protecting Lean Muscle Mass
To combat the potential for muscle loss when energy expenditure surges, we often look to growth hormone secretagogues. Compounds like Ipamorelin are highly favored during this phase. Ipamorelin stimulates the body's natural release of growth hormone without aggressively spiking cortisol or prolactin. Elevated growth hormone helps partition nutrients, favoring muscle preservation and further enhancing lipolysis (fat breakdown). It serves as the perfect "shield" for lean tissue while the triple-agonist does the heavy lifting on fat loss.
For those looking at mitochondrial optimization alongside muscle protection, MOTS-c is heavily researched for its ability to enhance exercise capacity and improve metabolic homeostasis at the cellular level. When the metabolism speeds up, having mitochondria that can efficiently handle that energy transfer is highly beneficial.
Gastrointestinal and Systemic Recovery
Any peptide that alters gastric emptying and gut transit times can cause occasional mild gastrointestinal distress, especially during dose titration. To soothe the gut lining and promote systemic anti-inflammatory recovery, integrating a BPC-157 protocol has become the gold standard among health enthusiasts. BPC-157 (Body Protection Compound) is renowned for its ability to accelerate tissue repair and heavily support digestive health, ensuring that the body absorbs nutrients efficiently even when appetite is low.
Comparative Analysis: The Peptide Tier List
To quickly visualize how adjusting your research model impacts bodily function, let's look at the differences between the three generations of weight-management peptides.
| Peptide Generation | Receptors Targeted | Primary Mechanism of Action | Energy/Metabolic Effect | Best Used For |
|---|---|---|---|---|
| Generation 1 (e.g., Semaglutide) | GLP-1 | Appetite suppression, slowed gastric emptying, insulin regulation. | Neutral to slightly lower (due to reduced food intake). | Initial weight loss, curbing food noise, basic metabolic resets. |
| Generation 2 (e.g., Tirzepatide) | GLP-1 + GIP | Enhanced appetite control, improved lipid handling, lowered nausea. | Neutral. The body utilizes fat better but overall basal burn doesn't spike. | Moderate to high weight loss, overcoming initial plateau. |
| Generation 3 (e.g., Retatrutide) | GLP-1 + GIP + Glucagon | Appetite control, aggressive liver fat clearance, triggered lipolysis. | High. Basal metabolic rate actively increases; acts as an internal furnace. | Breaking severe plateaus, advanced obesity models, aggressive body recomposition. |
Navigating Quality Control in Advanced Peptide Research
As the molecular complexity of peptides increases—from synthesizing a single chain to formulating a stable triple-hybrid molecule—the necessity for flawless quality control becomes absolutely paramount. You simply cannot transition to a highly advanced protocol using sub-par materials.
When glucagon, GIP, and GLP-1 receptors are engaged simultaneously, slight impurities or degradation in the peptide chain can lead to chaotic immune responses, injection site reactions, or highly diminished efficacy. At Alpha Carbon Labs, we recognize that the methodological requirements for these advanced research models place extreme demand on product purity.
We encourage all researchers and health optimization enthusiasts to rigorously review our quality control standards. Advanced amino acid sequences require stringent verification parameters, including HPLC (High-Performance Liquid Chromatography) and Mass Spectrometry, to guarantee that the molecular integrity remains intact. Before making any transition to advanced peptides, always verify the batch data via our COA documents (Certificates of Analysis).
Real-World Expectations: What Does the Transition Feel Like?
For individuals stepping beyond the dual-agonist ceiling, knowing what to physically expect makes the journey much smoother. Based on extensive research and user data, here is the typical timeline and sensation of upgrading to a triple-agonist:
- Weeks 1-2 (The Acclimation Phase): You may feel a slightly elevated core body temperature and perhaps an increase in resting heart rate by 5 to 10 beats per minute. This is entirely normal. It is the glucagon receptor revving up your body's internal engine. You will quickly notice you are sweating more during exercise. Appetite suppression is present but might feel "different" than the heavy, sometimes bloated feeling of single GLP-1s.
- Weeks 3-4 (The Mobilization Phase): The scale, which may have been stuck for months, typically begins to move again. At this phase, liver fat is rapidly breaking down. Users often report a surge in energy levels, contrasting sharply with the lethargy frequently experienced on older weight-loss medications.
- Weeks 5+ (The Recomposition Phase): The metabolism plateaus at a higher functional baseline. Subcutaneous and visceral fat decreases steadily. Because energy is higher, individuals often find they can push harder during physical training, facilitating better body recomposition (building muscle while shedding fat).
Answers to Common Questions (FAQ)
Is Retatrutide "better" than Tirzepatide?
It is not necessarily about being "better," but rather being a more powerful tool for specific scenarios. If you are early in your optimization journey, Tirzepatide is a brilliant, highly effective dual-agonist that will yield incredible results. Retatrutide is best viewed as the next logical step—the heavy hitter reserved for when your body has adapted and fights back against standard dual-agonist therapy.
Will the increased metabolic rate make me jittery?
Unlike classic thermogenic fat burners or stimulants (like caffeine or ephedrine) that heavily stimulate the central nervous system, the glucagon mechanism works on a cellular metabolic level, particularly in the liver. Most people do not experience "jitters." Instead, they report feeling a sustained warmth, smoother energy throughout the day, and increased thermogenesis (sweating) during physical activity.
Do I have to take it forever to keep the weight off?
This is the million-dollar question in metabolic optimization. Peptides are tools that correct metabolic dysfunction, clear liver fat, and reset insulin sensitivity. However, if you remove the tool and return to a lifestyle of processed foods, poor sleep, and inactivity, the weight will return. The optimal strategy is to use these peptides to clear the metabolic roadblocks, build lean muscle mass (which permanently raises your metabolic rate anyway), and establish sustainable routines that act as long-term anchors for your health.
Can I stack other performance peptides with a triple agonist?
Yes. As mentioned earlier in this guide, employing compounds aimed at muscle preservation (secretagogues) or tissue recovery (like BPC) is very common and heavily documented as highly synergistic in elite wellness circles.
Conclusion: Breaking Through the Ceiling
The science of human optimization is moving at a blistering pace. Just a few years ago, the idea of completely erasing "food noise" seemed like science fiction. Today, we are already moving past simple appetite suppression and venturing into a brand new era of active metabolic enhancement.
The dual-agonist ceiling is frustrating, but it is a natural biological defense mechanism. Trying to overcome it by starving yourself further will only ruin your metabolic baseline and destroy your hard-earned muscle. The methodological transition to a triple-agonist like Retatrutide respects evolutionary biology: rather than engaging in a war of restriction, it simply changes the hormonal instructions, telling the body to turn up the heat, burn via the liver, and utilize fuel like a much younger, entirely optimized machine.
By prioritizing hydration, protein intake, and precise dosing protocols, researchers and wellness seekers can safely harness the full spectrum of triple-agonism. The ceiling has been broken; all that is left is to optimize the journey upward.
References
- 1. Jastreboff, A. M., Kaplan, L. M., Frías, J. P., Wu, Q., Du, Y., Guler, S., ... & Coskun, T. (2023). Triple-Hormone-Receptor Agonist Retatrutide for Obesity—A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
- 2. Rosenstock, J., Frias, J., Jastreboff, A. M., Du, Y., Lou, J., Guler, S., ... & Haupt, A. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. The Lancet, 402(10401), 529-544.
- 3. Urva, S., Coskun, T., Haupt, A., et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, powerfully resolves liver fat in NAFLD. Journal of Hepatology.
- 4. Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., Wharton, S., Connery, L., Alves, B., ... & Stefanski, A. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine, 387(3), 205-216.
- 5. Wilding, J. P., Batterham, R. L., Calanna, S., Davies, M., Van Gaal, L. F., Lingvay, I., ... & Kushner, R. F. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine, 384(11), 989-1002.
- 6. Tschöp, M. H., Finan, B., Clemmensen, C., Gelfanov, V., Perez-Tilve, D., Hofmann, S. M., ... & DiMarchi, R. (2016). Unimolecular Polypharmacy for Treatment of Diabetes and Obesity. Cell Metabolism, 24(1), 51-62.
- 7. Finan, B., Clemmensen, C., Zhu, Z., Stemmer, K., Gauthier, K., Müller, L., ... & DiMarchi, R. D. (2015). Chemical hybridization of glucagon and thyroid hormone optimizes therapeutic impact for metabolic disease. Nature Medicine, 21(2), 223-228.
- 8. Müller, T. D., Blüher, M., Tschöp, M. H., & DiMarchi, R. D. (2022). Anti-obesity drug discovery: advances and challenges. Nature Reviews Drug Discovery, 21(3), 201-223.
- 9. Min, T., & Bain, S. C. (2021). The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Therapy, 12(1), 143-157.
- 10. Samocha-Bonet, D., Dixit, V. D., Kahn, C. R., et al. (2012). Metabolically active and inactive phases of weight loss and maintenance. Journal of Clinical Investigation.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.