The Enthesis Axis: Mechanistic Insights into F-Actin Remodeling with BPC-157 and TB-500 in Tendon-to-Bone Interface Models
Discover how the synergistic blend of BPC-157 and TB-500 accelerates stubborn joint and tendon recovery by supercharging cellular communication and F-actin scaffolding at the tendon-to-bone interface.
The Frustration of Stubborn Joint and Tendon Injuries
If you have ever dealt with a nagging joint injury, you already know the frustration. Whether it is a rotator cuff tear that keeps you up at night, Achilles tendonitis that derails your running routine, or a stubbornly painful case of tennis elbow, tendon injuries are notoriously slow to heal. For decades, the standard medical advice has been to rely on rest, ice, compression, and elevation—better known as the RICE method. Later, anti-inflammatory medications and physical therapy are thrown into the mix. Yet, despite our best efforts, these injuries often linger for months or even years.
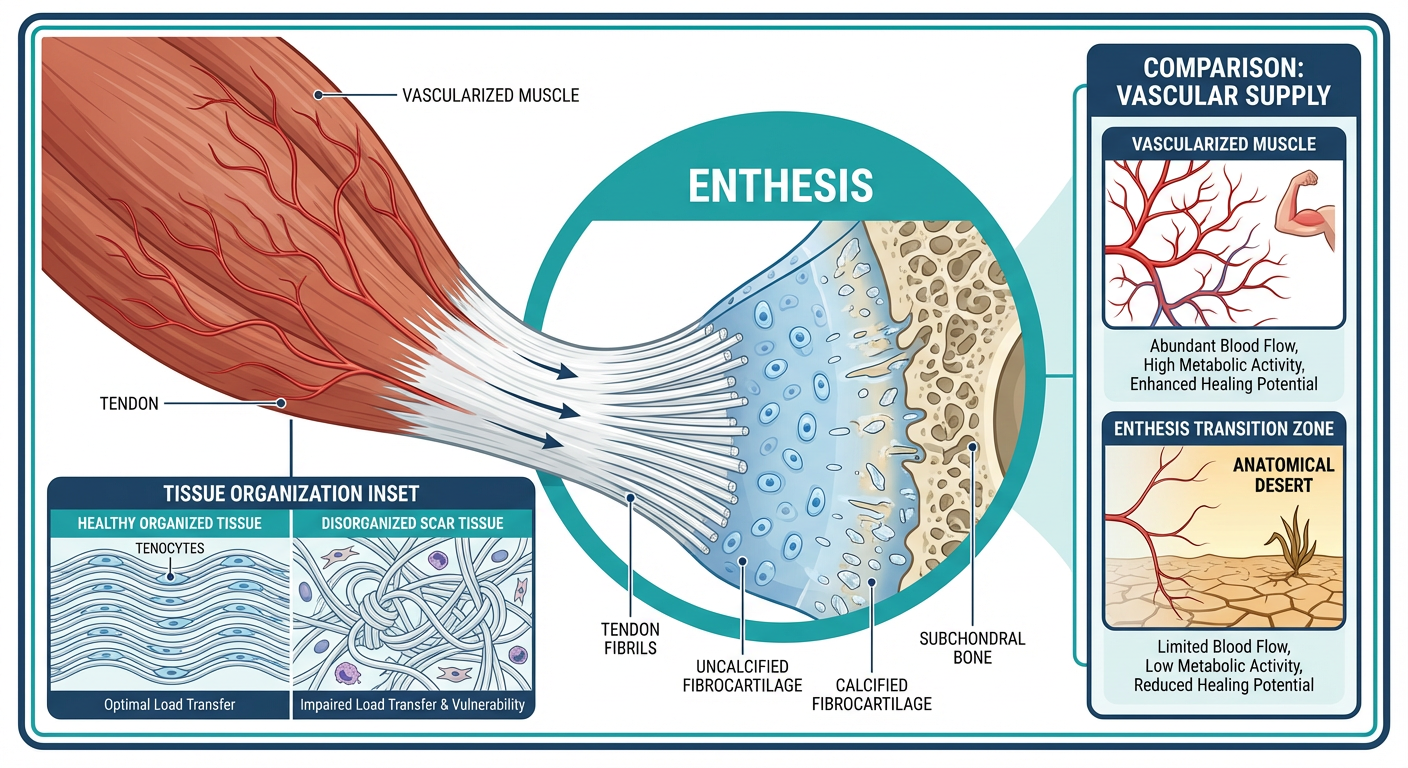
Why do these specific injuries take so long to heal compared to a muscle tear or a skin wound? The answer lies deep within your anatomy, in a highly specialized transition zone known to scientists and researchers as the enthesis. The enthesis is the exact micro-location where your flexible tendon tightly anchors into your rigid bone. It is a marvel of human biomechanics, designed to transfer massive amounts of physical force from your contracting muscles directly to your skeletal system, allowing you to run, lift, jump, and throw.
However, this structural marvel comes with a massive biological downside: it is a notorious weak link. The tendon-to-bone interface has very poor blood flow. When you injure a highly vascular area like a bicep muscle, fresh blood rushes to the scene, delivering oxygen, nutrients, and immune cells to rebuild the broken tissue rapidly. When you damage the enthesis, however, the area is practically an anatomical desert. Without sufficient blood flow, the natural tools required for repair simply cannot reach the injury site in meaningful quantities. As a result, the body often resorts to patching the area with unorganized scar tissue, leaving the anchor point weak, stiff, and highly prone to re-injury.
For health-conscious adults, functional athletes, and anyone dedicated to graceful anti-aging and mobility, accepting chronic joint pain is not an option. This is exactly why the wellness and optimization communities have turned their attention toward breakthrough research peptides. Today, we are exploring the profound mechanistic role of two of the most popular recovery compounds on the market: BPC-157 and TB-500. More specifically, we are going to unpack how the synergistic combination found in the BPC-157 + TB-500 Blend targets the cellular scaffolding of your joints, focusing on a critical regenerative process called F-Actin remodeling and cellular communication pathways like Focal Adhesion Kinase (FAK) signaling.
Do not let the scientific terminology intimidate you. By the end of this comprehensive guide, you will understand exactly how these peptides give your body the blueprint and the raw materials needed to rebuild your most stubborn injuries from the ground up.
Understanding the Enthesis: The Body's Biological Anchor
To truly grasp how targeted peptide therapy can accelerate healing, we first need to understand the battlefield. The term "enthesis" (plural: entheses) might sound like complex medical jargon, but it simply refers to the "insertion point" where connective tissues—like tendons and ligaments—attach to the bone. Think of it as the ultimate biological anchor. There are two primary types of entheses in the human body: fibrous and fibrocartilaginous.
Fibrous entheses generally occur on the long shafts of bones, where the tendon attaches directly. However, the fibrocartilaginous entheses—found in the Achilles tendon, the rotator cuff in the shoulder, and the patellar tendon in the knee—are the ones that cause the most trouble. These transition zones feature four distinct layers: pure tendon, uncalcified fibrocartilage, calcified fibrocartilage, and finally, bone. This gradual shift in tissue type is a brilliant evolutionary design meant to act as a shock absorber. If the flexible tendon attached abruptly to the hard, unyielding bone, the sheer mechanical stress of daily movement would cause it to snap immediately.
But the complex multi-layered structure of the fibrocartilaginous enthesis makes it a nightmare to repair once damaged. When a micro-tear occurs in this zone—often due to repetitive stress, mechanical overload, or the natural decline in collagen production associated with aging—it triggers a cascade of localized inflammation. In a healthy scenario, the body would clear out the damaged cells and lay down fresh, organized collagen fibers to seamlessly bridge the gap.
Unfortunately, because this zone lacks a dense network of blood vessels (avascularity), the cellular repair signals are incredibly weak. The body adopts a "quick fix" strategy. Instead of carefully regenerating the specialized four-layer shock absorber, it haphazardly dumps Type III collagen—which is structurally inferior and highly disorganized—into the gap. This creates fibrotic scar tissue. Scar tissue is rigid, non-elastic, and creates a "weakest link" scenario in the kinetic chain. Every time you try to return to the gym or the tennis court, that stiff scar tissue pulls painfully against the surrounding healthy tissue, creating chronic inflammation and eventually tearing all over again.
This endless cycle of micro-tearing and poor healing at the enthesis is exactly what we refer to as tendinopathy. Beating tendinopathy requires more than just masking the pain with ibuprofen or resting until it "feels okay." It requires a fundamental shift in how the damaged cells behave at a microscopic level. It requires enhanced blood flow to deliver supplies, and a massive upregulation in the cellular scaffolding process known as F-actin remodeling. This is where advanced therapeutic options leapfrog traditional recovery methods.
Cellular Scaffolding: The Magic of F-Actin Remodeling Explained
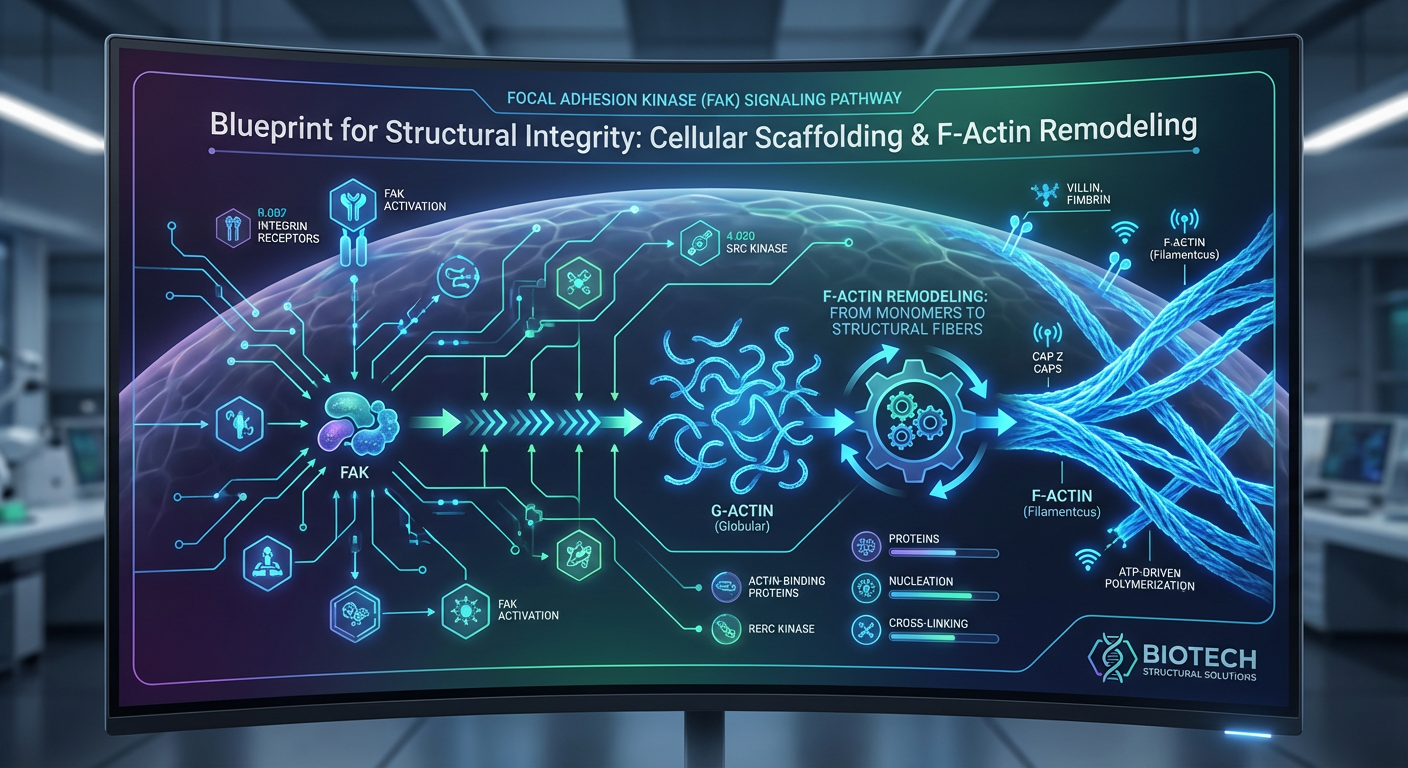
If we zoom in on the tendon-to-bone interface during the healing process, we are essentially looking at a microscopic construction site. And just like any construction site, before you can pour concrete or build walls, you need a sturdy internal framework. For human cells—especially the specialized tendon cells called tenocytes, and the builder cells called fibroblasts—that internal framework is composed of a protein called actin. Actin makes up the cytoskeleton, which is literally the cell's skeleton.
Actin exists primarily in two forms within our cells. The first form is G-actin (Globular actin), which you can think of as individual thousands of loose Lego bricks floating around inside the cell, waiting to be used. The second form is F-actin (Filamentous actin). When a cell gets the signal that it needs to move, stretch, or repair tissue, it quickly snaps all those loose G-actin Lego bricks together to form long, extremely strong chains called F-actin.
This dynamic process of snapping these proteins together and taking them apart is what scientists call F-actin remodeling. Why is F-actin remodeling so absolutely critical for healing joint injuries?
When you sustain a tear at the enthesis, your fibroblasts and tenocytes need to migrate—literally crawl—across the gap of the injury to lay down new, healthy collagen. Cells do not have legs; they move by continuously extending their internal F-actin filaments forward, gripping the surrounding tissue, and pulling the rest of the cell body behind them. Furthermore, F-actin filaments structurally reinforce the new tissue matrix, allowing the cells to withstand mechanical tension as the tendon heals.
If your F-actin remodeling process is slow or impaired due to age, poor blood flow, or chronic inflammation, the builder cells physically cannot migrate to the tear fast enough to repair it properly. The construction site stalls. The tissue remains weak and degraded. This is the hidden root cause of why your tennis elbow has not improved in six months. Optimizing F-actin remodeling is the biological equivalent of giving your repair cells high-speed transport and infinite scaffolding materials. And amazingly, certain peptide strings naturally occurring in the body are specifically designed to regulate and supercharge this exact mechanism.
FAK Signaling: The Foreman of the Cellular Construction Site
Having plenty of F-actin available to build cellular scaffolding is wonderful, but the cells still need a director to tell them exactly where to go and what to do when they get there. Enter Focal Adhesion Kinase, or FAK. FAK signaling is one of the most important communication pathways in connective tissue regeneration.
Let's stick with our construction analogy. If the tenocytes and fibroblasts are the construction workers, and F-actin is the scaffolding they use to climb and build, then FAK is the site foreman. FAK is an enzyme that gets activated through mechanical tension and chemical signals. When activated, FAK acts as a massive communication hub. It relays messages from the outside environment of the cell directly into the cell's nucleus, essentially shouting, "We have sustained mechanical damage here! Start migrating, begin F-actin remodeling, and lay down fresh Type I collagen immediately!"
Research has shown that FAK activation is vital for healthy tendon-to-bone healing. When FAK signaling is robust, cells stick together beautifully (adhesion), move precisely to where they are needed (migration), and proliferate rapidly to close the tear. When FAK signaling is weak—which is highly common in the avascular enthesis zone—the cells sit dormant, and the injury lingers indefinitely.
Medical researchers have spent years trying to figure out how to upregulate FAK signaling and accelerate F-actin remodeling simultaneously, safely, and naturally. Standard pharmaceutical anti-inflammatories like NSAIDs (ibuprofen, naproxen) actually inhibit natural healing cascades by blunting the initial signaling required to start the repair process. The ultimate goal has always been to find a compound that supports the body's native healing pathways without simply suppressing symptoms. This ongoing quest illuminated the immense restorative power of biologically active peptides.
BPC-157: The Master of Angiogenesis and Cellular Signaling
Now that we understand the core problems preventing tendon healing—lack of blood flow, suppressed FAK signaling, and stalled F-actin remodeling—we can look at a the solutions. The first half of the ultimate recovery equation is Body Protection Compound 157, universally known as BPC-157.
BPC-157 is a sequence of 15 amino acids that is derived from a protective protein naturally found in the human gastric tract. In the stomach, its primary role is to rapidly repair ulcers, protect the digestive lining from harsh stomach acid, and promote rapid cellular turnover. However, researchers quickly discovered that when this identical peptide string is administered systemically or locally near an injury, its healing properties extend far beyond the digestive system.
BPC-157 is quite literally a master regenerative signal. It tackles the challenge of repairing the enthesis from two powerful angles:
1. Triggering Massive Angiogenesis
Remember how the tendon-to-bone connection is an avascular desert that is starved for bold flow? BPC-157 acts as an oasis. It is a potent stimulator of a protein called Vascular Endothelial Growth Factor (VEGF). By severely upregulating VEGF, BPC-157 commands the body to undergo angiogenesis—the creation of brand-new, microscopic blood vessels.
These tiny new capillaries push their way into the previously avascular enthesis, establishing direct supply lines to the injured tissue. For the first time, your chronically injured tendon is flushed with nutrient-rich, highly oxygenated blood, dramatically shifting the environment from stagnant and inflamed to active and regenerative.
2. Amplifying FAK and Paxillin Signaling
Beyond simply improving logistics via blood flow, BPC-157 acts directly on the cellular communication hubs. Scientific studies involving tendon fibroblasts exposed to BPC-157 have demonstrated a remarkable phenomenon: the peptide significantly increases the expression and activation of Focal Adhesion Kinase (FAK) and another associated protein called paxillin.
In simple terms, BPC-157 arms the "cellular foreman" with a megaphone. The FAK signals become incredibly loud and clear. The tenocytes suddenly recognize the damage, adhere strongly to the injury site, and begin migrating at an accelerated rate to stitch the micro-tears back together. By forcing the cells out of a dormant state and into an active repair cycle, BPC-157 ensures the body is actively trying to heal the root cause of the discomfort rather than just mitigating the inflammation.
TB-500: The Ultimate Architect of F-Actin Structuring
While BPC-157 sets up the supply chain and shouts the initial orders through FAK signaling, it needs a partner to actually execute the mass remodeling of the cellular scaffolding at maximum speed. This is where TB-500 steps onto the stage as the ultimate cellular architect.
TB-500 is a synthetic fraction of a naturally occurring, larger protein called Thymosin Beta-4. Thymosin Beta-4 is present in almost every human cell, but it is heavily concentrated in wound fluid, blood platelets, and white blood cells—the first responders to any injury. Its dominant biological purpose is almost exclusively related to managing actin inside our cells.
If BPC-157 tells the cell when and where to move, TB-500 gives the cell the physical ability to move.
Direct F-Actin Modulation
TB-500's magic lies in its role as an "actin-sequestering" molecule. When an injury occurs, TB-500 essentially grabs hold of those loose G-actin Lego bricks we talked about earlier. It regulates their assembly into the long, powerful F-actin chains. Without enough Thymosin Beta-4 activity, F-actin chains become rigid, or they don't form quickly enough, paralyzing the cell's ability to migrate effectively to the enthesis tear.
By heavily supplementing the environment with TB-500, you are drastically increasing the efficiency of F-actin remodeling. The tenocytes and fibroblasts become incredibly flexible, dynamic, and mobile. They can literally stretch out further and crawl faster across the damaged tendon-to-bone interface. This rapid migration means the structural void in the tendon is filled much faster.
Fighting Fibrosis and Scar Tissue
Because TB-500 speeds up the cellular migration process so effectively, the body doesn't need to resort to its "quick fix" backup plan of dumping rigid Type III collagen (scar tissue) into the tear. Instead, the well-supported fibroblasts are able to carefully lay down aligned, highly flexible Type I collagen fibers. This is the difference between an Achilles tendon that heals properly, allowing you to run smoothly for years to come, and one that heals with a big knot of scar tissue that constantly aches every cold morning.
The Perfect Synergy: Why Combine BPC-157 and TB-500?
Understanding the individual mechanisms of these two peptides makes it immediately apparent why they are rarely discussed in isolation anymore. When it comes to conquering profound tendinopathies or recovering from connective tissue surgeries, using them simultaneously attacks the problem from every conceivable biological angle.
- The BPC-157 Contribution: It forces the growth of new blood vessels into the dead-zone of the joint (angiogenesis) and triggers the FAK communication pathway so the cells know it's time to work.
- The TB-500 Contribution: It naturally binds to actin inside the cells, radically accelerating the F-actin remodeling process so the cells can physically execute the repair work in record time.
This synergistic cross-talk transforms a dormant, painful, non-healing injury into a highly active regeneration zone. For those who want the absolute gold standard in comprehensive tissue recovery, integrating these perfectly paired modes of action is an unmatched approach. For individuals experiencing severe, multi-faceted tissue breakdown, some advanced protocols even introduce heavy-hitting, copper-binding compounds into the mix to further regulate collagen synthesis. You can explore the expanded three-part synergy available in options like the BPC-157 + TB-500 + GHK-Cu Blend for profound restorative effects on both internal joints and external superficial tissues.
The Three Stages of Peptide-Assisted Healing
Let's map out exactly what taking control of your recovery via the enthesis axis looks like. The biological healing of connective tissue happens in three distinct phases mathematically mapped by modern physiology. Introducing the synergistic BPC/TB blend severely condenses the timeline of these phases.
1. The Inflammatory Phase (Days 1 to 5)
Traditionally, this phase is characterized by intense swelling, heat, and pain as immune cells rush the area to clear debris. While inflammation is necessary to kick off healing, excessive inflammation causes secondary damage to healthy cells. BPC-157 modulates this response. It does not blindly crush inflammation like a steroid; rather, it upregulates protective mechanisms to cap the inflammatory damage while immediately initiating the FAK signals to transition into the next phase. Pain relief is often remarkably fast during this window.
2. The Proliferative Phase (Weeks 1 to 6)
This is where the magic of F-actin remodeling takes over. In a normal body, a severe rotator cuff tear might stall in this phase for months because the cells are too slow to migrate. With TB-500 heavily present in the system, fibroblast migration goes into overdrive. BPC-157's newly formed capillaries pump in the necessary oxygen. New, flexible collagen networks are rapidly woven across the bone-to-tendon gap.
3. The Remodeling Phase (Months 2 to 12+)
Even after the pain is gone, the body spends up to a year realigning the new collagen fibers so they can handle heavy mechanical loads (like deadlifts, sprinting, or throwing a baseball). The continued expression of FAK, supported by sustained cellular signaling, ensures that the structural integrity of the enthesis matches the pre-injury state as closely as possible, vastly reducing the chance of snapping the tendon again.
Beyond the Blend: Optimizing Your Recovery Ecosystem
While the combination of BPC-157 and TB-500 is arguably the most potent direct intervention for tendon, cartilage, and bone repair, a holistic approach to healing forces you to consider systemic wellness. Peptides are messengers; they tell your body to perform vital processes. But your body still requires raw energy, profound restorative sleep, and nutritional building blocks to carry out those orders.
For individuals recovering from massive traumas—such as a bicep tear surgery or complex ligament reconstruction—layering complementary growth factors can elevate recovery even higher. Peptides that mimic the regenerative pulse of youthful growth hormone, such as IGF-1LR3 (Insulin-like Growth Factor 1 Long Arg3), play a staggering role in muscle cell hyperplasia and overall tissue repair. When IGF-1LR3 ensures the surrounding muscle tissue stays anabolic and robust, it takes the sheer mechanical strain off the healing tendon, giving the BPC/TB sequence a much higher rate of success at the enthesis.
Furthermore, maintaining adequate hydration, optimizing protein intake (with high concentrations of glycine and proline for collagen production), prioritizing 8 hours of sleep, and performing gentle, controlled physical therapy loads will naturally stimulate the mechanical tension needed to keep FAK signaling healthy. Remember, you must gently load the tendon to tell the new collagen which direction to orient itself. Rest alone is rust.
The Crucial Importance of Sourcing and Quality Control
When dealing with precision cellular signaling, purity is not just a preference; it is mandatory. The F-actin remodeling and FAK cascading effects discussed in this article were discovered in clinical and laboratory models utilizing high-purity, structurally intact peptide sequences. If a peptide is poorly synthesized, degraded, or contaminated with heavy metals or byproducts, it will utterly fail to bind to the correct cellular receptors. At best, a low-quality peptide will do nothing; at worst, it can trigger unwanted immune responses.
For consumers seeking real biological optimization, verifying the integrity of your research protocols is the first step toward success. Always seek out suppliers that prioritize transparency. Understanding how therapeutic compounds are produced via modern peptide synthesis provides insight into why careful manufacturing matters. Moreover, uncompromising quality control ensures that the delicate amino acid sequences of BPC-157 and TB-500 remain strictly stable from the lab exactly to your doorstep. You should never compromise on biological research inputs; always verify the molecular weight and purity by checking the supplier's third-party COA documents (Certificates of Analysis).
Comparing Peptide Therapy to Traditional Healing Methods
To summarize just how vast the gap is between standard care and peptide-assisted tissue regeneration, let's visualize the differences side-by-side.
| Mechanism/Outcome | Traditional RICE & NSAIDs Method | BPC-157 & TB-500 Synergy Method |
|---|---|---|
| Inflammation Handling | Blunts all inflammation, inadvertently stalling the initial repair signals necessary for healing. | Modulates inflammation gracefully while upregulating cellular communication (FAK) to begin immediate repair. |
| Blood Flow (Angiogenesis) | Ice severely restricts blood flow, furthering the avascular nutrient starvation of the enthesis. | BPC-157 triggers VEGF, actively growing new capillary vessels directly into the injury site. |
| Cellular Migration | Slow and unassisted. Healing is delayed for months or years in stubborn areas. | TB-500 heavily drives F-actin remodeling, allowing fibroblasts to sprint to the injury zone. |
| Tissue Quality Outcome | Often results in fibrotic scar tissue (Type III collagen) that is stiff and prone to re-tearing. | Promotes organized, flexible Type I collagen networks for robust, highly elastic mechanical load bearing. |
Frequently Asked Questions (FAQ)
How does the BPC-157 and TB-500 blend target different types of joint pain?
Because the blend fundamentally operates at a cellular level—by rebuilding actual scaffolding and growing fresh blood vessels—it does not discriminate between an Achilles tear, a tennis elbow, or patellar tendonitis. As long as the injury falls within the connective tissue, muscle, or gut domain, the upregulated FAK signaling and F-actin remodeling pathways will inherently target that damaged tissue to promote systemic recovery.
Is it necessary to use them together, or can I just use one?
While you can absolutely utilize BPC-157 or TB-500 individually with fantastic results, using them together is universally considered the gold standard for severe or stubborn injuries. They act on entirely different but complementary pathways. BPC-157 improves local blood flow and boosts biological "start signals" (FAK), while TB-500 acts directly on the proteins that give the cell flexibility and speed (F-actin). Together, they are the complete package.
How long does it typically take to notice improvements in stubborn tendon issues?
While biological response times vary wildly depending on age, nutrition, and injury severity, many individuals report significant reductions in nagging pain and systemic inflammation within the first two weeks of research. However, for true, structural enthesis rebuilding—where the collagen fibers actually re-anchor securely to the bone—research suggests allowing a minimum of 6 to 12 weeks of sustained cellular support.
Will resolving F-actin remodeling help prevent future injuries?
Yes. The central problem with traditional healing is the formation of brittle scar tissue. By utilizing peptides to ensure the body heals using highly organized, flexible Type I collagen (facilitated by proper F-actin formation and migration), the newly healed tissue is much closer to its original, youthful elasticity. This deeply minimizes the risk of suffering another tear in the exact same spot.
Are there any specific lifestyle habits that enhance the peptide recovery protocol?
Absolutely. Tendon healing is highly responsive to mechanical loading. Complete, prolonged bed rest is counterproductive. Light, eccentric physical therapy movements encourage the active FAK pathways to lay the newly remodeled collagen perfectly along the lines of mechanical stress. Nutrition is also paramount—ensure sufficient vitamin C, zinc, protein, and hydration, which are the physical building blocks your cells use once the peptides instruct them to start building.
Final Thoughts: Taking Control of the Enthesis Axis
Navigating the grueling timeline of joint, tendon, and ligament injuries can severely impact an active individual's quality of life. The old paradigms of waiting around for an avascular zone to heal itself are frustrating, slow, and frequently result in permanent biomechanical weaknesses.
Through modern scientific advancements, we now have an intimate understanding of the microscopic battleground at the tendon-to-bone interface. By specifically targeting the foundational cellular scaffolding—focusing heavily on F-actin remodeling to speed up construction and FAK signaling to broadcast clear repair instructions—we can fundamentally change the environment of an injury. The combination of BPC-157's incredible capacity to forge new blood vessels with TB-500's elegant control over internal cell flexibility creates a biological synergy that traditional medicines simply cannot match.
Whether you are desperately trying to rehab a blown rotator cuff, finally cure that lingering medial epicondylitis in your elbow, or looking to structurally bulletproof your body as you age gracefully, unlocking the secrets of the enthesis axis is your key to sustained, pain-free mobility.
References
- 1. Chang CH, Tsai WC, Hsu YH, Pang JH. The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration. Journal of Applied Physiology. 2011;110(3):774-780.
- 2. Kesić V, et al. Gastric pentadecapeptide BPC 157 in tendon healing. European Journal of Pharmacology. 2016;789:223-229.
- 3. Philp D, Goldstein AL, Kleinman HK. Thymosin beta4 promotes angiogenesis, wound healing, and hair follicle development. Mechanisms of Ageing and Development. 2004;125(2):113-115.
- 4. Sosne G, Qiu P, Kurpakus-Wheater M, Matthew C. Thymosin beta 4: tissue repair and regeneration. Vitamins and Hormones. 2010;82:205-227.
- 5. Thomopoulos S, Genin GM, Galatz LM. The development and function of the tendon-to-bone attachment. Annual Review of Biomedical Engineering. 2010;12:67-90.
- 6. Goldstein AL, Hannappel E, Kleinman HK. Thymosin beta4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine. 2005;11(9):421-429.
- 7. Schaller MD. Cellular functions of FAK kinases: insight into molecular mechanisms and novel functions. Journal of Cell Science. 2010;123(Pt 7):1007-1013.
- 8. Vukojevic J, et al. Pentadecapeptide BPC 157 and the angiogenesis in tissue healing. Current Pharmaceutical Design. 2018;24(18):1990-2001.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.