Telomeric Integrity: Evaluating NAD+ Influence on Replicative Senescence in In-Vitro Models
This comprehensive analysis explores the biochemical mechanisms linking NAD+ depletion to telomere shortening and replicative senescence. We evaluate in-vitro evidence regarding the NAD+-Sirtuin-Telomere axis and compare metabolic interventions with peptide bioregulators like Epithalon.
The Convergence of Metabolic Flux and Genomic Stability
The intricate relationship between metabolic cofactors and genomic stability represents one of the most promising frontiers in longevity research. Specifically, the interplay between Nicotinamide Adenine Dinucleotide (NAD+) levels and telomere maintenance offers a mechanistic explanation for the onset of replicative senescence. For researchers exploring the therapeutic potential of metabolic intervention, understanding how NAD+ influences the shelterin complex, sirtuin activity, and DNA repair mechanisms is paramount.
Telomeres, the repetitive TTAGGG nucleotide sequences capping the ends of eukaryotic chromosomes, serve as the "mitotic clock" of the cell. With each division, the "end replication problem" dictates that telomeres shorten, eventually triggering a DNA Damage Response (DDR) that leads to cell cycle arrest—a state known as senescence. However, contemporary data suggests that this process is not merely a fixed mechanical countdown but is heavily influenced by the metabolic environment of the nucleus.
This article evaluates the impact of NAD+ availability on maintaining telomeric integrity in in-vitro models, analyzing the biochemical pathways through which NAD+ depletion accelerates senescence and how restoration may mitigate genomic instability. Furthermore, we will contextualize these findings alongside established peptide bioregulators such as Epithalon and energetic modulators like SS-31 and MOTS-c.
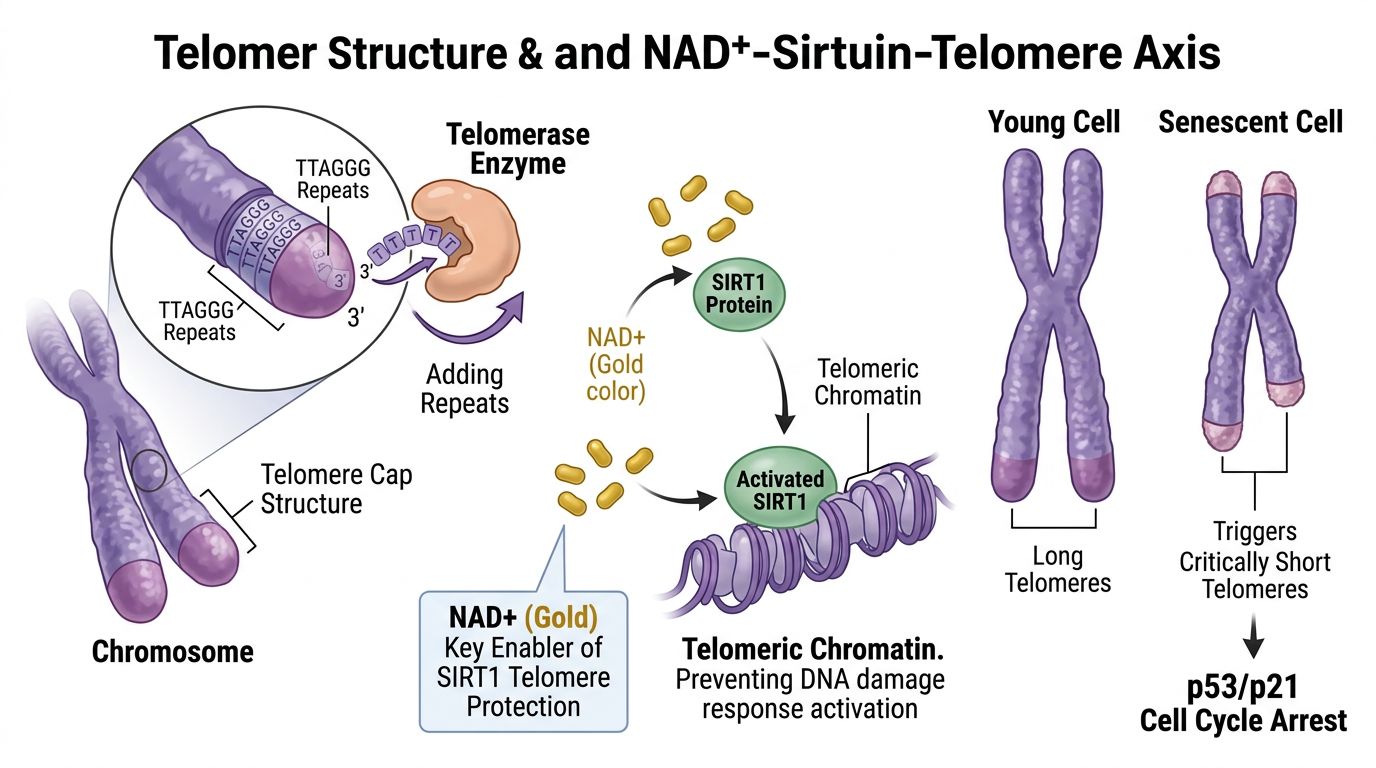
The NAD+-Sirtuin-Telomere Axis
The central hypothesis connecting NAD+ to telomere length relies on the activity of Sirtuins (Silent Information Regulator 2 proteins), particularly SIRT1 and SIRT6. These Class III histone deacetylases are obligatory NAD+-dependent enzymes; unlike their zinc-dependent counterparts, sirtuins cannot function without adequate NAD+ bioavailability. Consequently, intracellular NAD+ concentration acts as the rate-limiting substrate for sirtuin activity.
SIRT1: The Guardian of Telomeric Chromatin
SIRT1 is perhaps the most extensively studied sirtuin in the context of genomic stability. In the nucleus, SIRT1 aids in the maintenance of constitutive heterochromatin. Research indicates that SIRT1 positive regulation leads to the deacetylation of histone H3 lysine 9 (H3K9) and H4 lysine 16 (H4K16) at telomeric regions.
This deacetylation compacts the chromatin, stabilizing the telomere and preventing fragility. When NAD+ levels decline—a hallmark of aging observed across mammalian species—SIRT1 activity plummets. This loss of deacetylation leads to a more "open" chromatin structure at the telomeres, increasing their susceptibility to recombination events and nuclease attacks.
SIRT6: The DNA Repair Coordinator
SIRT6 is often dubbed the "longevity sirtuin" due to its critical role in DNA Double-Strand Break (DSB) repair. Telomeres, by their nature, resemble DSBs to the cell's repair machinery. The shelterin complex prevents the cell from "fixing" these ends, which would result in catastrophic chromosome fusion. However, actual damage to the telomeric DNA requires precise repair.
SIRT6 is recruited to sites of DNA damage in an NAD+-dependent manner. Once there, it stimulates the polymerization of PARP1 (Poly [ADP-ribose] polymerase 1) and recruitment of repair factors. Crucially, SIRT6 is required for the stable association of telomeric chromatin with the nuclear envelope, a positioning vital for proper replication. In SIRT6-deficient models (mimicking low NAD+ states), telomeres exhibit dysfunction characterized by end-to-end chromosomal fusions and premature senescence.
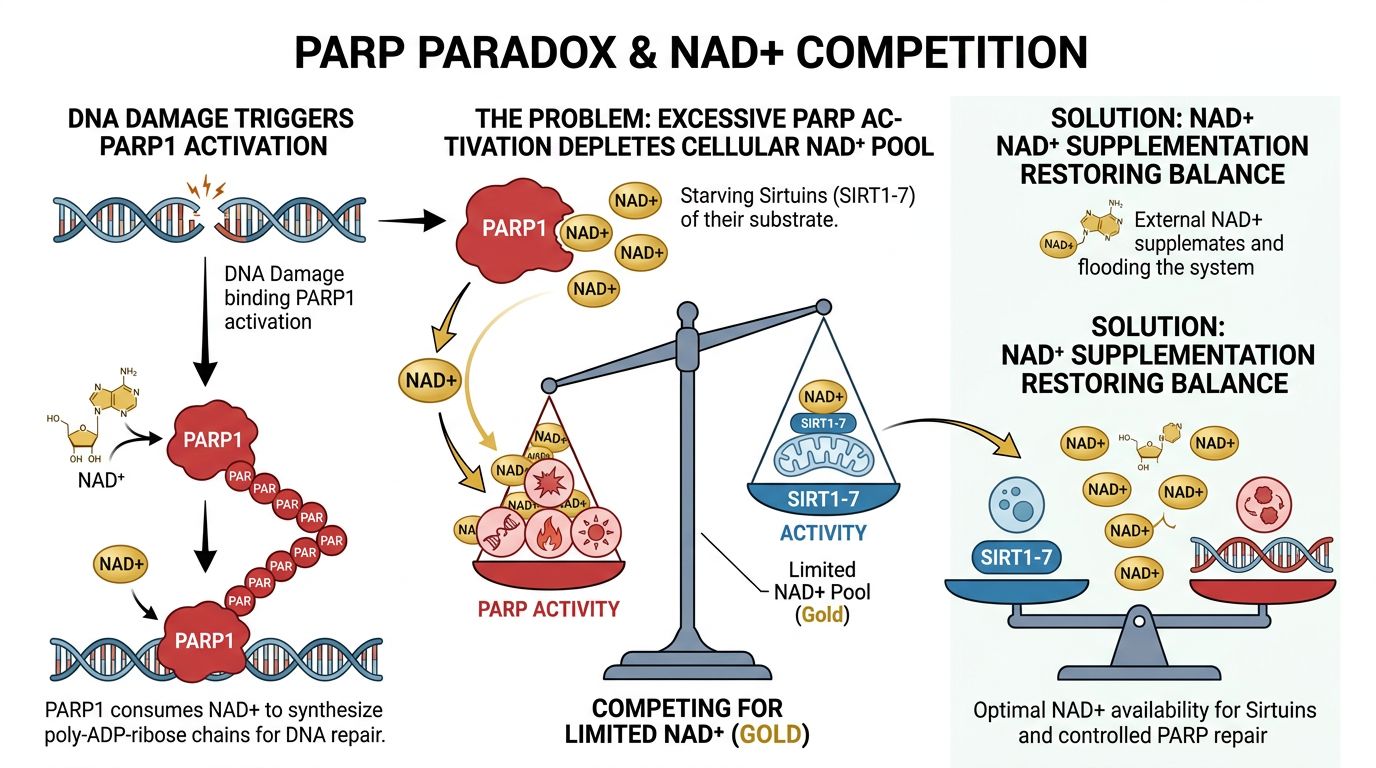
The PARP Paradox: Competition for the NAD+ Pool
Understanding telomeric integrity requires an analysis of the "tug-of-war" for NAD+ resources between Sirtuins and PARPs. PARP1 is a DNA repair enzyme that detects single-strand breaks. Upon activation, PARP1 consumes massive amounts of NAD+ to synthesize poly-ADP-ribose chains.
In the context of replicative senescence, cells often accumulate low-level DNA damage. This chronic damage leads to constitutive PARP1 hyperactivation, which drastically depletes the intracellular NAD+ pool. The consequence is a metabolic crisis:
- DNA damage triggers PARP1.
- PARP1 lowers NAD+ concentrations.
- Low NAD+ starves SIRT1 and SIRT6.
- Lack of Sirtuin activity leads to telomere de-protection and further genomic instability.
- Senescence is accelerated.
Supplementation with NAD+ in in-vitro cultures has been shown to break this cycle. By replenishing the pool, researchers observe that even in the presence of DNA damage, Sirtuins can remain active enough to maintain telomeric heterochromatin, essentially buying the cell time to repair itself without entering permanent arrest.
In-Vitro Evidence: Reversing the Senescence Phenotype
Recent studies utilizing fibroblasts and Hematopoietic Stem Cells (HSCs) provide compelling evidence for the efficacy of NAD+ repletion. In experiments specifically measuring telomere length via quantitative fluorescence in situ hybridization (Q-FISH), cells treated with NAD+ precursors or direct NAD+ exhibited maintained telomere length over more population doublings compared to controls.
The PGC-1α and TERT Connection
A landmark study by Amano et al. identified a critical feedback loop involving p53. Telomere dysfunction activates p53, which is known to arrest cell growth. However, p53 also binds to the promoters of PGC-1α (a master regulator of mitochondrial biogenesis), repressing it. This suppression causes mitochondrial dysfunction, leading to ROS (Reactive Oxygen Species) generation, which further damages telomeres.
The introduction of NAD+ intervenes by activating SIRT1. SIRT1 deacetylates p53, reducing its ability to repress PGC-1α. Furthermore, SIRT1 is suggested to modulate the expression of TERT (Telomerase Reverse Transcriptase) in certain cell types. This suggests that NAD+ does not merely protect existing telomeres but may create a permissive environment for telomerase activity where it is naturally expressed.
| Molecular Pathway | Role of NAD+ | Consequence of NAD+ Depletion | Outcome of NAD+ Restoration |
|---|---|---|---|
| SIRT1 Deacetylation | Substrate for enzyme | Telomeric chromatin opens (fragile) | Heterochromatin compaction (stable) |
| PARP1 Activity | Substrate for enzyme | Competition limits Sirtuin function | Simultaneous repair and regulation allowed |
| Mitochondrial ROS | SIRT3 cofactor | High ROS damages telomeric DNA | SIRT3 reduces ROS, sparing telomeres |
| Telomerase (TERT) | Indirect regulator via Epigenetics | Downregulation or inhibited access | Improved accessibility and activity |
Comparative Analysis: NAD+ vs. Telomeric Peptides
For researchers designing comprehensive anti-senescence protocols, it is essential to distinguish between the foundational support provided by NAD+ and the specific signaling actions of peptides like Epithalon. While both arguably support "anti-aging" via telomeres, their mechanisms of action are distinct and potentially synergistic.
Epithalon: Direct Telomerase Activation
Epithalon (Ala-Glu-Asp-Gly) is a synthetic tetrapeptide modeled after a pineal gland extract. Its primary mechanism regarding senescence is the upregulation of telomerase gene expression. Unlike NAD+, which creates the supportive environment for genomic stability, Epithalon acts as a specific signal to elongate telomeres.
Research indicates that Epithalon can induce telomerase activity in somatic cells that typically have low or undetectable activity. This makes it a direct intervention for telomere elongation, whereas NAD+ is a broad-spectrum intervention for telomere protection and maintenance.
Mitochondrial Synergy: SS-31 and MOTS-c
Since telomere integrity is intimately linked to mitochondrial health (via the ROS axis described above), combining NAD+ research with mitochondrial-derived peptides offers a multi-vector approach.
- SS-31: This peptide targets the inner mitochondrial membrane, specifically interacting with cardiolipin. By stabilizing the electron transport chain, SS-31 reduces ROS production at the source. This reduces the oxidative burden on telomeres, complementing the repair capacity boosted by widespread NAD+ availability.
- MOTS-c: A mitochondrial-derived peptide that regulates metabolic homeostasis. Similar to NAD+, MOTS-c mimics a state of exercise or fasting, activating AMPK. The concurrent use of NAD+ and MOTS-c could theoretically act synergistically to enhance the p53/PGC-1α axis, further protecting cells from senescence induced by metabolic dysregulation.
The Role of GHK-Cu in DNA Repair
While discussing genomic integrity, the tripeptide GHK-Cu warrants mention. While GHK-Cu is primarily known for tissue remodeling and collagen synthesis, transcriptomic studies reveal it modulates the expression of numerous genes involved in DNA repair and antioxidant defense.
In an in-vitro model of senescence, GHK-Cu has been shown to restore the "stemness" of cells and reduce inflammatory markers (SASP - Senescence Associated Secretory Phenotype). A research protocol combining the metabolic fuel of NAD+ with the gene-modulating effects of GHK-Cu presents a robust model for studying cellular reversion.
Methodological Considerations: Purity and Synthesis
When conducting research on replicative senescence, the purity of the input variables is critical. The degradation of NAD+ in solution can yield nicotinamide, which is actually a Sirtuin inhibitor. Therefore, using high-purity, lyophilized NAD+ that is reconstituted immediately prior to use is essential for valid data.
Similarly, the efficacy of peptides like Epithalon or SS-31 relies on proper peptide synthesis to ensure correct amino acid sequencing and the absence of trifluoroacetic acid (TFA) salts that can be cytotoxic to delicate cell cultures. Researchers should refer to quality control documentation and liquid chromatography-mass spectrometry (LC-MS) reports to verify the integrity of their reagents.
Future Directions in Senescence Research
The evaluation of telometic integrity via NAD+ modulation represents a shift from viewing aging as wear-and-tear to viewing it as a programmable metabolic decline. Current research is expanding into "super-cocktails" involving BPC-157 for cytoprotection alongside NAD+ and Epithalon.
Furthermore, the emergence of newer agents such as FOXO4-DRI (a senolytic) suggests a two-step future for therapy: first, clearing senescent cells, and second, restoring the NAD+/Telomere axis in the remaining healthy population to prevent further decline.
Conclusion
In-vitro models confirm that NAD+ is not merely a passive energy carrier but a decisive regulator of telomere length and stability. By fueling Sirtuins and satisfying the hunger of PARPs during DNA repair, NAD+ helps maintain the "protective cap" of the genome. While peptides like Epithalon offer direct telomerase activation, NAD+ ensures the chromatin landscape is conducive to stability. For the advanced researcher, the manipulation of the NAD+ metabolome offers a foundational strategy for interrogating the mechanisms of cellular aging.
For detailed information on reagent purity and synthesis standards, please review our peptide synthesis protocols and COA documents.
References
- 1. Amano, H., et al. (2019). Telomere Dysfunction Induces Sirtuin Repression that Drives Telomere-Dependent Disease. Cell Metabolism, 29(6), 1274-1290.
- 2. Sahin, E., & Depinho, R. A. (2010). Linking functional decline of telomeres, mitochondria and stem cells during ageing. Nature, 464, 520–528.
- 3. Imai, S., & Guarente, L. (2014). NAD+ and sirtuins in aging and disease. Trends in Cell Biology, 24(8), 464-471.
- 4. Covarrubias, A. J., et al. (2021). NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology, 22, 119–141.
- 5. Palacios, J. A., et al. (2010). SIRT1 contributes to telomere maintenance and augments global homologous recombination. Journal of Cell Biology, 191(7), 1299–1313.
- 6. Khavinson, V. K., et al. (2003). Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bulletin of Experimental Biology and Medicine, 135(6), 590-592.
- 7. Tennen, R. I., et al. (2011). SIRT6 is required for maintenance of telomere position effect in human cells. Nature Communications, 2, 433.
- 8. Fang, E. F., et al. (2016). NAD+ Replenishment Improves Lifespan and Healthspan in Ataxia Telangiectasia Models via Mitophagy and DNA Repair. Cell Metabolism, 24(4), 566-581.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.