Retatrutide vs. Tirzepatide vs. Semaglutide — What the Research Shows
A comprehensive, data-driven comparison of three incretin-based peptides: their receptor profiles, clinical trial outcomes, mechanisms of action, and what published research reveals about their differences.
Introduction: The Incretin Revolution in Metabolic Research
The landscape of metabolic peptide research has undergone a dramatic transformation over the past two decades. At the center of this shift are three compounds that have generated enormous scientific and clinical interest: Semaglutide, Tirzepatide, and Retatrutide. Each represents a different generation and strategy in the evolution of incretin-based therapeutics, and together they illustrate how receptor pharmacology, molecular engineering, and clinical trial design have converged to produce some of the most impactful findings in modern obesity and diabetes research.
This article provides a comprehensive, side-by-side analysis of these three peptides based exclusively on published clinical trial data, peer-reviewed research, and regulatory documents. The goal is not to advocate for any compound, but to clearly present what the science shows—mechanism of action, receptor binding profiles, pharmacokinetic properties, efficacy data from major clinical trials, safety profiles, and emerging research directions. For researchers and scientists evaluating these compounds, understanding the distinctions between a single GLP-1 receptor agonist, a dual GIP/GLP-1 agonist, and a triple GIP/GLP-1/glucagon agonist is essential for designing rigorous studies and interpreting results.
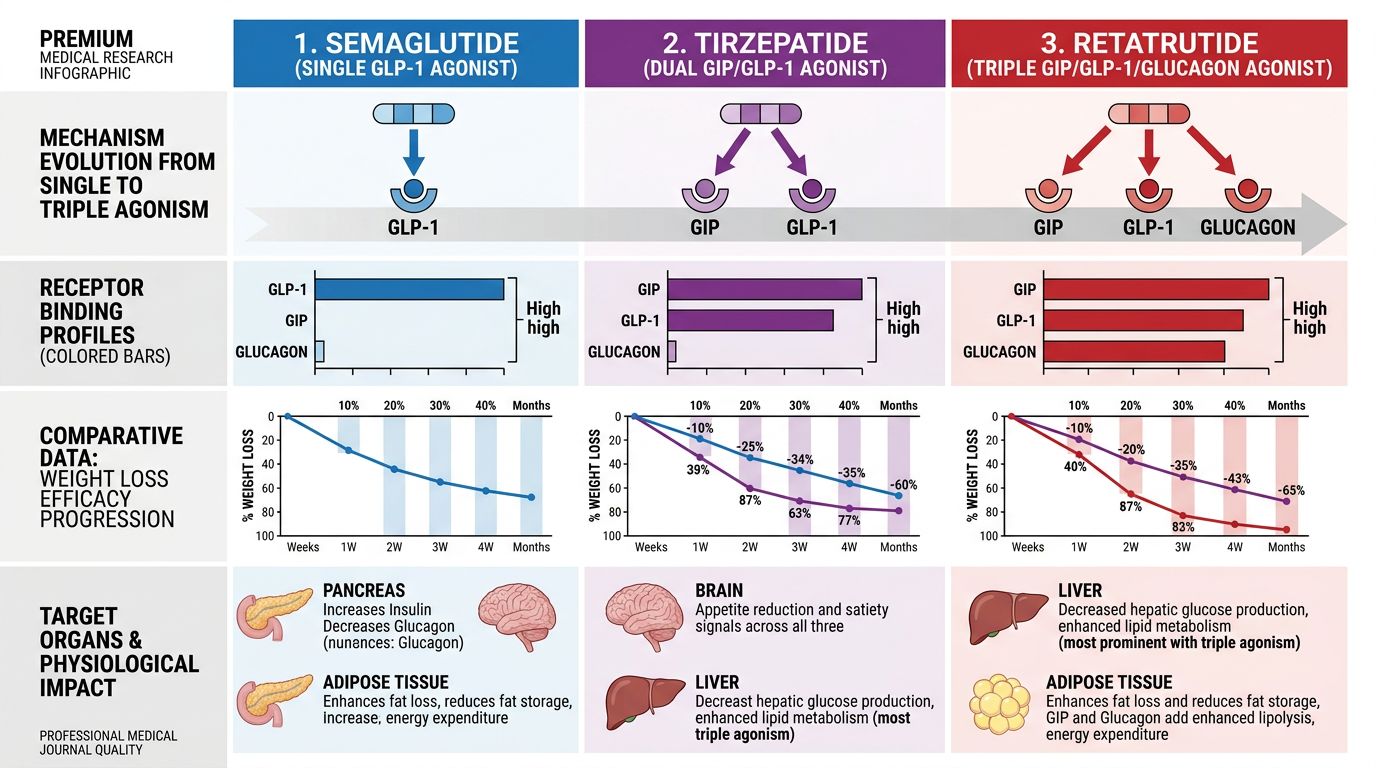
Mechanisms of Action: From Single to Triple Agonism
Semaglutide — Selective GLP-1 Receptor Agonism
Semaglutide is a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist. It was developed by Novo Nordisk as a modified analog of native human GLP-1, with specific amino acid substitutions and a fatty acid side chain (C-18 fatty diacid) that enable binding to serum albumin. This albumin binding dramatically extends its half-life to approximately 165 hours (~7 days), allowing once-weekly subcutaneous administration. The key amino acid substitutions include an Aib (alpha-aminoisobutyric acid) at position 8, which confers resistance to degradation by the enzyme dipeptidyl peptidase-4 (DPP-4), and an arginine at position 34.
GLP-1 is an endogenous incretin hormone secreted by intestinal L-cells in response to nutrient ingestion. It acts on GLP-1 receptors expressed in the pancreatic beta cells, the gastrointestinal tract, the central nervous system (particularly the hypothalamus, brainstem, and area postrema), and the cardiovascular system. Through these receptors, GLP-1 agonists like Semaglutide stimulate glucose-dependent insulin secretion, suppress glucagon release, slow gastric emptying, and reduce appetite via central satiety pathways. Research has demonstrated that semaglutide crosses the blood-brain barrier and directly activates GLP-1 receptors in the arcuate nucleus and other hypothalamic regions involved in energy homeostasis (Gabery et al., 2020).
Tirzepatide — Dual GIP/GLP-1 Receptor Agonism
Tirzepatide, developed by Eli Lilly, is a first-in-class dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist. Its molecular design is based on the native GIP sequence with modifications that enable simultaneous activation of both the GIP receptor (GIPR) and the GLP-1 receptor (GLP-1R). The peptide incorporates a C-20 fatty diacid moiety attached via a linker, which facilitates albumin binding and extends the half-life to approximately 5 days, also enabling once-weekly dosing.
The rationale for dual agonism is rooted in the observation that GIP and GLP-1 are the two primary incretin hormones, together accounting for approximately 50–70% of postprandial insulin secretion. While GLP-1 receptor agonism had been extensively validated, the role of GIP in metabolic regulation was historically controversial. Early research suggested GIP signaling was impaired in type 2 diabetes, leading some to question its therapeutic utility. However, preclinical work by Finan et al. (2013) demonstrated that unimolecular dual agonists targeting both GIPR and GLP-1R produced superior glycemic and weight outcomes compared to selective GLP-1 agonism alone—a finding that laid the groundwork for tirzepatide's development.
Tirzepatide has approximately 5-fold greater potency at the GIP receptor relative to the GLP-1 receptor. Research suggests this imbalanced agonism may be mechanistically important: GIP receptor activation in adipose tissue appears to enhance lipid buffering capacity and improve insulin sensitivity, while GIP signaling in the brain may complement GLP-1-mediated appetite suppression through distinct neural circuits (Samms et al., 2020). The dual mechanism thus engages complementary metabolic pathways not accessible through GLP-1 agonism alone.
Retatrutide — Triple GIP/GLP-1/Glucagon Receptor Agonism
Retatrutide (LY3437943), also developed by Eli Lilly, represents the next evolutionary step: a triple agonist that simultaneously activates the GIP receptor, the GLP-1 receptor, and the glucagon receptor (GCGR). This unimolecular peptide integrates three distinct receptor-binding activities into a single compound, a feat of significant molecular engineering. Like tirzepatide, it uses a C-20 fatty diacid for albumin binding and once-weekly dosing, with a half-life of approximately 6 days.
The addition of glucagon receptor agonism is the critical differentiator. Glucagon, traditionally viewed as a counter-regulatory hormone that raises blood glucose, has emerged in research as a potent activator of hepatic energy expenditure, lipid oxidation, and thermogenesis. Preclinical studies by Day et al. (2009) and Pocai et al. (2009) demonstrated that co-agonism at the glucagon receptor, when balanced by GLP-1-mediated insulin secretion, could produce net weight loss without hyperglycemia—effectively harnessing glucagon's catabolic effects while maintaining glycemic safety through incretin activity.
Retatrutide was specifically engineered with balanced activity at all three receptors. Research by Coskun et al. (2022) characterized its in vitro pharmacology, showing potent activation of human GIPR, GLP-1R, and GCGR in cell-based assays. The glucagon component is hypothesized to increase resting energy expenditure, promote hepatic fatty acid oxidation, reduce hepatic lipid content, and potentially activate brown adipose tissue thermogenesis—mechanisms not engaged by GLP-1 or GIP agonism alone.
Receptor Binding Profile Comparison
Understanding the receptor pharmacology of these three peptides is essential for interpreting their differential effects. The following table summarizes their receptor activity:
| Property | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| GLP-1 Receptor | Full agonist (primary target) | Agonist (secondary to GIP) | Agonist (one of three targets) |
| GIP Receptor | None | Full agonist (primary target, ~5x potency vs GLP-1R) | Full agonist |
| Glucagon Receptor | None | None | Full agonist |
| Half-life | ~165 hours (~7 days) | ~5 days | ~6 days |
| Dosing | Once weekly (SC) | Once weekly (SC) | Once weekly (SC) |
| Albumin Binding | C-18 fatty diacid | C-20 fatty diacid | C-20 fatty diacid |
| Developer | Novo Nordisk | Eli Lilly | Eli Lilly |
This progressive expansion of receptor targets—from one (GLP-1) to two (GIP + GLP-1) to three (GIP + GLP-1 + glucagon)—represents a deliberate strategy to engage increasingly comprehensive metabolic control. Each additional receptor adds a distinct physiological lever: GLP-1 for appetite suppression and glucose-dependent insulin release, GIP for incretin amplification and adipose tissue function, and glucagon for energy expenditure and hepatic lipid metabolism.
Clinical Trial Data: Weight Reduction
Semaglutide — The STEP Program
The Semaglutide Treatment Effect in People with Obesity (STEP) clinical trial program is one of the most extensive trial programs ever conducted for a weight management compound. The key trials and their results include:
STEP 1 (Wilding et al., NEJM, 2021): A randomized, double-blind, placebo-controlled trial enrolling 1,961 adults with BMI ≥30 (or ≥27 with comorbidities) without diabetes. Participants received semaglutide 2.4 mg weekly or placebo, plus lifestyle intervention, for 68 weeks. Results showed a mean body weight change of −14.9% with semaglutide vs. −2.4% with placebo (estimated treatment difference: −12.4 percentage points). Notably, 86.4% of semaglutide-treated participants achieved ≥5% weight loss, and 32.0% achieved ≥20% weight loss.
STEP 2 (Davies et al., Lancet, 2021): Conducted in 1,210 adults with type 2 diabetes and BMI ≥27. Semaglutide 2.4 mg produced −9.6% mean weight reduction vs. −3.4% for placebo at 68 weeks. This trial was significant because weight loss is typically more difficult to achieve in the diabetic population due to insulin resistance and concomitant medications.
STEP 3 (Wadden et al., JAMA, 2021): Combined semaglutide 2.4 mg with intensive behavioral therapy (30 counseling sessions). Mean weight loss was −16.0% with semaglutide vs. −5.7% with placebo, demonstrating that combining pharmacotherapy with structured lifestyle intervention enhances outcomes.
STEP 5 (Garvey et al., Nature Medicine, 2022): A 104-week trial demonstrating sustained efficacy. At 2 years, semaglutide 2.4 mg produced −15.2% weight loss vs. −2.6% for placebo, with maintained metabolic improvements in blood pressure, lipids, and HbA1c.
Tirzepatide — The SURMOUNT Program
Tirzepatide's weight management efficacy was evaluated in the SURMOUNT clinical trial program, which produced landmark results:
SURMOUNT-1 (Jastreboff et al., NEJM, 2022): A pivotal trial enrolling 2,539 adults with BMI ≥30 (or ≥27 with comorbidities) without diabetes. Participants were randomized to tirzepatide 5 mg, 10 mg, or 15 mg weekly, or placebo, for 72 weeks. Results were striking: mean weight reductions of −15.0% (5 mg), −19.5% (10 mg), and −20.9% (15 mg) vs. −3.1% for placebo. At the highest dose, 36.2% of participants achieved ≥25% weight loss—an unprecedented result in a Phase III obesity trial.
SURMOUNT-2 (Garvey et al., Lancet, 2023): Conducted in 938 adults with type 2 diabetes and BMI ≥27. Tirzepatide 10 mg and 15 mg produced mean weight reductions of −12.8% and −14.7%, respectively, vs. −3.2% for placebo at 72 weeks. These results exceeded those of semaglutide 2.4 mg in the diabetic population (STEP 2: −9.6%).
SURMOUNT-3 (Wadden et al., 2023): Evaluated tirzepatide following an intensive lifestyle intervention lead-in period. After 12 weeks of diet/exercise producing ~6% initial weight loss, randomization to tirzepatide resulted in total weight loss of −26.6% (max dose) vs. −5.0% for placebo at 72 weeks, demonstrating additive benefit on top of lifestyle modification.
Retatrutide — Phase II Trial
Retatrutide's clinical data comes from its Phase II dose-finding trial, published in the New England Journal of Medicine (Jastreboff et al., NEJM, 2023). This trial enrolled 338 adults with BMI ≥30 (or ≥27 with comorbidities) and tested multiple doses (1, 4, 8, and 12 mg weekly) against placebo for 48 weeks.
The results were remarkable: at the 12 mg dose, participants achieved a mean weight reduction of −24.2% at 48 weeks. Importantly, the weight loss trajectory had not plateaued at 48 weeks, suggesting that longer treatment duration could produce even greater reductions. At the 12 mg dose, 26% of participants lost more than 30% of their body weight. These numbers exceeded anything previously reported for any single-agent pharmacotherapy for obesity at a comparable timepoint.
It must be emphasized that this is Phase II data with smaller sample sizes compared to the Phase III STEP and SURMOUNT programs. Phase III trials for retatrutide (the TRIUMPH program) are underway and will provide the definitive large-scale efficacy and safety data needed for regulatory evaluation.
Comparative Efficacy Summary
| Metric | Semaglutide 2.4 mg | Tirzepatide 15 mg | Retatrutide 12 mg |
|---|---|---|---|
| Trial Phase | Phase III (STEP) | Phase III (SURMOUNT) | Phase II |
| Population | Non-diabetic obesity | Non-diabetic obesity | Non-diabetic obesity |
| Duration | 68 weeks | 72 weeks | 48 weeks |
| Mean Weight Loss (%) | −14.9% | −20.9% | −24.2% |
| ≥5% Weight Loss | 86.4% | 91.0% | 93% |
| ≥10% Weight Loss | 69.1% | 79.5% | 83% |
| ≥20% Weight Loss | 32.0% | 36.2% | 63% |
These cross-trial comparisons must be interpreted with caution. Differences in trial design, duration, patient populations, lifestyle interventions, and sample sizes preclude direct head-to-head conclusions. Only randomized controlled trials directly comparing these agents can definitively establish superiority. Nevertheless, the pattern across trials is consistent with the hypothesis that engaging additional metabolic receptors produces incrementally greater weight reduction.
Glycemic Control: HbA1c Reductions
Semaglutide in Type 2 Diabetes (SUSTAIN Program)
Semaglutide's glycemic efficacy was established in the SUSTAIN trial program for type 2 diabetes. SUSTAIN 1–5 demonstrated HbA1c reductions of 1.5–1.8 percentage points with semaglutide 1.0 mg weekly, consistently superior to active comparators including sitagliptin, exenatide ER, and insulin glargine. In SUSTAIN 6 (Marso et al., NEJM, 2016), semaglutide also demonstrated cardiovascular safety with a significant 26% reduction in major adverse cardiovascular events (MACE), marking an important milestone for GLP-1 agonist research.
Tirzepatide in Type 2 Diabetes (SURPASS Program)
Tirzepatide's glycemic effects were evaluated in the SURPASS trial program. In SURPASS-2 (Frías et al., NEJM, 2021), a head-to-head comparison against semaglutide 1.0 mg in 1,879 adults with type 2 diabetes, tirzepatide at all doses (5, 10, and 15 mg) demonstrated statistically superior HbA1c reductions: −2.01%, −2.24%, and −2.30% for tirzepatide vs. −1.86% for semaglutide. At the 15 mg dose, 51.7% of participants achieved HbA1c <5.7% (normal range), compared to 19.7% with semaglutide 1.0 mg. This is the only large-scale, direct head-to-head comparison between these compounds.
Retatrutide in Type 2 Diabetes
In the Phase II trial, Retatrutide demonstrated HbA1c reductions of up to −2.02% at the highest doses in participants with type 2 diabetes. Notably, 71% of participants receiving retatrutide 12 mg achieved HbA1c <5.7%. While promising, these results await confirmation in larger Phase III trials. The addition of glucagon receptor agonism, which would theoretically raise glucose levels, did not produce net hyperglycemia—supporting the hypothesis that concurrent GLP-1 and GIP agonism provides sufficient glycemic counterbalance.
Safety and Tolerability Profiles
Gastrointestinal Side Effects
All three compounds share a common class effect of gastrointestinal adverse events, primarily nausea, vomiting, diarrhea, and constipation. These effects are attributable to GLP-1 receptor activation in the gastrointestinal tract and central nervous system, and they are generally dose-dependent and most prominent during dose escalation.
In the STEP trials, Semaglutide 2.4 mg was associated with nausea in 44.2% of participants (vs. 17.4% placebo), diarrhea in 31.5%, and vomiting in 24.8%. Most events were mild-to-moderate and transient, typically resolving within the first 8–12 weeks of treatment. The discontinuation rate due to adverse events was 7.0% for semaglutide vs. 3.1% for placebo.
In SURMOUNT-1, Tirzepatide 15 mg produced nausea in 31.0%, diarrhea in 23.0%, and vomiting in 12.2%. Discontinuation rates due to adverse events ranged from 4.3% (5 mg) to 7.1% (15 mg). The GI tolerability profile of tirzepatide appeared modestly better than semaglutide in absolute terms, though direct comparison requires the SURPASS-2 head-to-head data, where GI event rates were broadly similar between tirzepatide 15 mg and semaglutide 1.0 mg.
In the Phase II trial, Retatrutide showed GI side effects consistent with the class: nausea (varying by dose from 16% to 46%), diarrhea (14–35%), and vomiting (9–23%). The overall tolerability was considered acceptable by investigators, with a low discontinuation rate due to adverse events (approximately 6% across dose groups). The addition of glucagon receptor agonism did not appear to introduce a qualitatively different GI side effect profile compared to dual or single agonists.
Cardiovascular Safety
Cardiovascular outcomes are a critical consideration for any metabolic therapy. Semaglutide has the most robust cardiovascular data. SUSTAIN 6 demonstrated a statistically significant 26% reduction in MACE (composite of cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke) compared to placebo. The SELECT trial (Lincoff et al., NEJM, 2023) further confirmed a 20% reduction in MACE with semaglutide 2.4 mg in overweight/obese adults without diabetes, establishing cardiovascular benefit beyond glycemic control.
Tirzepatide's cardiovascular outcomes trial (SURPASS-CVOT) is ongoing, with results expected in 2025–2026. Pre-specified meta-analysis of Phase III data has not shown cardiovascular safety signals, and tirzepatide has demonstrated significant improvements in cardiovascular risk factors including blood pressure, triglycerides, and inflammatory markers.
Retatrutide does not yet have dedicated cardiovascular outcomes data. However, in the Phase II trial, retatrutide produced significant reductions in systolic blood pressure, triglycerides, and other cardiometabolic risk markers. The glucagon receptor component is of particular interest because preclinical data suggest glucagon may have direct cardioprotective effects through enhanced myocardial fatty acid oxidation and improved cardiac energetics (Berglund et al., 2023).
Hepatic Effects and MASLD/NASH
A notable area of differentiation is the effect on hepatic steatosis and metabolic dysfunction-associated steatotic liver disease (MASLD, formerly NAFLD/NASH). The glucagon receptor component of Retatrutide has direct hepatic effects that distinguish it from the other two compounds.
In the Phase II trial, retatrutide produced dramatic reductions in liver fat content as measured by MRI-proton density fat fraction (MRI-PDFF). At 48 weeks, the 12 mg dose group showed a mean relative reduction in liver fat of approximately 82–86%, with the majority of participants achieving normalization of liver fat content (<5% PDFF). These results significantly exceeded those reported for semaglutide and tirzepatide in comparable imaging substudies.
Semaglutide has shown meaningful reductions in liver fat (e.g., ~30% relative reduction in some studies), and a Phase IIb trial for NASH showed histological improvement. Tirzepatide demonstrated a 52.1% relative reduction in liver fat at 52 weeks in the SYNERGY-NASH trial. While these are significant, retatrutide's magnitude of liver fat reduction—attributable to glucagon-mediated hepatic lipid oxidation—appears substantially greater, positioning it as a particularly interesting compound for MASLD/NASH research.
Pharmacokinetic Considerations
All three compounds share the design principle of fatty acid-mediated albumin binding for half-life extension, enabling once-weekly subcutaneous dosing. However, their pharmacokinetic profiles differ in ways that may be clinically relevant:
Semaglutide has the longest half-life (~165 hours), resulting in very stable steady-state plasma concentrations with low peak-to-trough ratios. This pharmacokinetic stability may contribute to its consistent efficacy and the relatively predictable GI tolerability profile during steady state. Semaglutide achieves steady state after approximately 4–5 weeks of once-weekly dosing.
Tirzepatide's half-life of approximately 5 days is somewhat shorter but still supports effective once-weekly dosing. Its pharmacokinetics are dose-proportional across the 5–15 mg range, and it reaches steady state in approximately 4 weeks.
Retatrutide's half-life of approximately 6 days falls between the other two compounds. Population pharmacokinetic analysis from the Phase II trial indicated linear pharmacokinetics across the dose range tested, with steady-state achieved by approximately week 4 of the dose-escalation schedule.
Body Composition Effects
A critical question in weight management research is the composition of weight lost—specifically, the ratio of fat mass to lean mass reduction. Ideally, weight loss should preferentially reduce adipose tissue while preserving lean body mass (skeletal muscle).
DEXA substudy data from the STEP program showed that approximately 60–65% of weight lost with Semaglutide was fat mass, with 35–40% being lean mass. This ratio is consistent with historical data for dietary weight loss, suggesting that semaglutide does not preferentially spare or deplete lean tissue.
Tirzepatide showed a potentially more favorable body composition profile in SURMOUNT-1 substudy analyses. Approximately 67–70% of weight lost was fat mass, with 30–33% lean mass. Researchers have hypothesized that GIP receptor activation in adipose tissue may improve fat mobilization and adipose tissue insulin sensitivity, potentially leading to more favorable fat-to-lean loss ratios (Samms et al., 2022).
Retatrutide body composition data from the Phase II trial are limited but preliminary analyses suggest a similar or potentially more favorable fat-to-lean ratio, possibly due to glucagon-mediated increases in energy expenditure and lipid oxidation that preferentially target stored fat. Full DEXA analyses from Phase III trials will be needed to confirm this hypothesis.
Energy Expenditure and Metabolic Rate
One of the most significant theoretical advantages of including glucagon receptor agonism in Retatrutide's mechanism is its potential impact on energy expenditure. A common challenge with significant weight loss is the adaptive reduction in resting metabolic rate (metabolic adaptation), which can slow further weight loss and promote weight regain.
GLP-1 agonism (as with Semaglutide) primarily reduces energy intake through appetite suppression but does not appear to meaningfully increase energy expenditure. Some studies have suggested that semaglutide may even produce modest reductions in resting metabolic rate proportional to weight lost, consistent with metabolic adaptation.
GIP receptor agonism (engaged by both Tirzepatide and Retatrutide) has been associated with enhanced lipid buffering in white adipose tissue and potentially increased brown adipose tissue activity in preclinical models, though the magnitude of energy expenditure effects in humans remains uncertain.
Glucagon receptor agonism (unique to Retatrutide) is the most potent known pharmacological stimulator of hepatic energy expenditure. Glucagon activates hepatic fatty acid oxidation via AMPK and PPARα signaling, increases hepatic thermogenesis, and has been shown in preclinical models to increase whole-body energy expenditure by 10–15% (Habegger et al., 2010). If these effects translate to humans, they could partially offset metabolic adaptation and contribute to the greater weight loss observed with retatrutide. This hypothesis awaits confirmation from indirect calorimetry studies in Phase III trials.
Ongoing and Future Research
Semaglutide
Semaglutide has the most mature clinical program and the broadest range of approved indications. Current research directions include:
- Heart failure with preserved ejection fraction (HFpEF) — The STEP-HFpEF trial demonstrated significant improvements in symptoms, physical limitations, and exercise function in obese patients with HFpEF (Kosiborod et al., NEJM, 2023).
- Chronic kidney disease — The FLOW trial showed a 24% reduction in kidney disease progression.
- Alcohol use disorder and substance addiction — Early-stage trials are investigating GLP-1 effects on reward pathways.
- Oral semaglutide at higher doses (25 mg and 50 mg) for improved convenience.
- Combination with other agents (e.g., cagrilintide in CagriSema) for enhanced efficacy.
Tirzepatide
Tirzepatide research is expanding rapidly across multiple therapeutic areas:
- Obstructive sleep apnea (OSA) — The SURMOUNT-OSA trial demonstrated significant reduction in apnea-hypopnea index (AHI) scores.
- MASH/NASH — The SYNERGY-NASH trial showed histological improvement in steatohepatitis.
- Cardiovascular outcomes — The SURPASS-CVOT trial is ongoing.
- Heart failure — Trials evaluating tirzepatide in HFpEF and HFrEF are underway.
- Combination with Cagrilintide analogs for further weight management enhancement.
Retatrutide
Retatrutide's research pipeline is the earliest-stage but potentially the most transformative:
- Phase III TRIUMPH obesity program — Large-scale pivotal trials underway to confirm Phase II results.
- Phase III type 2 diabetes program — Evaluating glycemic efficacy and safety at scale.
- MASLD/NASH — Dedicated trials leveraging retatrutide's hepatic effects via glucagon receptor agonism.
- Cardiovascular outcomes — Planned or in development.
- Potential applications in lipodystrophy, metabolic syndrome, and polycystic ovary syndrome (PCOS) based on the broad metabolic activity profile.
Molecular Engineering: Design Principles
The three compounds illustrate an evolution in peptide engineering approaches:
Semaglutide represents the optimization of a single-target approach. By extensively modifying native GLP-1 (amino acid substitutions + fatty acid acylation), researchers created a molecule with dramatically improved pharmacokinetics while retaining selective receptor specificity. The success of this approach validated long-acting incretin agonism as a viable strategy for chronic metabolic disease management.
Tirzepatide represents the integration approach: building a single molecule that simultaneously engages two related but distinct receptors. This required careful optimization of the peptide sequence to achieve desired affinity ratios at GIPR and GLP-1R, balancing efficacy at each target while maintaining suitable pharmacokinetics. The unimolecular design (vs. co-administration of two separate molecules) ensures coordinated receptor activation and simplified dosing.
Retatrutide extends this to three targets, requiring even more sophisticated molecular design to achieve balanced activity at GIPR, GLP-1R, and GCGR. The challenge of activating the glucagon receptor—which would ordinarily raise blood glucose—while simultaneously engaging incretins to maintain glycemic safety, represents a precise pharmacological balancing act validated by the Phase II clinical data.
Research Implications and Study Design Considerations
For researchers designing studies involving these compounds, several considerations are important:
Dose-response relationships: All three compounds show clear dose-dependent effects on both efficacy outcomes and adverse events. Studies should consider titration schedules that balance tolerability with target exposure. The dose-escalation protocols used in clinical trials (typically starting at the lowest dose and increasing every 4 weeks) reflect the importance of GI tract adaptation.
Duration of treatment: Weight loss with these compounds generally continues for 40–72+ weeks before plateauing. Short-term studies may significantly underestimate maximal efficacy. This is particularly relevant for Retatrutide, where the 48-week Phase II data showed ongoing weight loss at study end.
Metabolic biomarkers: Beyond weight and HbA1c, researchers should consider hepatic fat content (MRI-PDFF), inflammatory markers (hsCRP, IL-6), lipid panels (including apolipoprotein B and lipoprotein(a)), body composition (DEXA), and energy expenditure (indirect calorimetry) to fully characterize metabolic effects.
Comparator selection: The choice of comparator has important implications. Placebo-controlled designs establish absolute efficacy but do not address comparative effectiveness. Active comparator trials (like SURPASS-2) are necessary but expensive and complex. Researchers should carefully consider whether their study design addresses clinically relevant questions about relative efficacy.
Conclusion: The Evolving Landscape of Incretin Research
The progression from Semaglutide to Tirzepatide to Retatrutide illustrates a broader principle in metabolic research: that targeting multiple complementary pathways can produce effects greater than the sum of individual components. Each compound has contributed uniquely to scientific understanding:
Semaglutide established that GLP-1 receptor agonism, optimized for pharmacokinetics and delivered at adequate doses, could produce clinically meaningful weight loss and cardiovascular benefit—changing the paradigm for pharmacological obesity treatment and opening the door to an entirely new therapeutic class.
Tirzepatide demonstrated that dual incretin agonism (GIP + GLP-1) could surpass selective GLP-1 agonism, challenging the conventional wisdom that GIP signaling was not therapeutically useful and revealing the importance of multi-receptor engagement for metabolic outcomes.
Retatrutide's Phase II data suggest that adding glucagon receptor agonism further amplifies metabolic effects—particularly on hepatic lipid content and potentially on energy expenditure—while maintaining glycemic safety through incretin co-agonism. If Phase III results confirm these findings, retatrutide could represent another step change in the field.
The research community is witnessing a period of unprecedented innovation in incretin-based metabolic science. As Phase III data mature for the newer compounds, head-to-head trials are conducted, and longer-term safety data accumulate, the relative positioning of these peptides will become clearer. What is already evident is that the understanding of multi-receptor pharmacology, peptide engineering, and metabolic physiology has advanced dramatically—driven in large part by the clinical development of these three remarkable compounds.
All peptides discussed in this article are available from Alpha Carbon Labs for research purposes only. These products are not intended for human consumption and are sold exclusively for legitimate scientific research.
References
- 1. Wilding, J. P. H., et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine, 384(11), 989-1002.
- 2. Jastreboff, A. M., et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine, 387(3), 205-216.
- 3. Jastreboff, A. M., et al. (2023). Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
- 4. Frías, J. P., et al. (2021). Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine, 385(6), 503-515.
- 5. Davies, M., et al. (2021). Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2). The Lancet, 397(10278), 971-984.
- 6. Marso, S. P., et al. (2016). Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine, 375(19), 1834-1844.
- 7. Finan, B., et al. (2013). Unimolecular Dual Incretins Maximize Metabolic Benefits in Rodents, Monkeys, and Humans. Science Translational Medicine, 5(209), 209ra151.
- 8. Coskun, T., et al. (2022). LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss. Cell Metabolism, 34(9), 1234-1247.
- 9. Gabery, S., et al. (2020). Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight, 5(6), e133429.
- 10. Samms, R. J., et al. (2020). GIPR agonism mediates weight-independent insulin sensitization by tirzepatide in obese mice. Journal of Clinical Investigation, 130(11), 5861-5876.
- 11. Lincoff, A. M., et al. (2023). Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. New England Journal of Medicine, 389(24), 2221-2232.
- 12. Garvey, W. T., et al. (2022). Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nature Medicine, 28(10), 2083-2091.
- 13. Garvey, W. T., et al. (2023). Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2). The Lancet, 402(10402), 613-626.
- 14. Kosiborod, M. N., et al. (2023). Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. New England Journal of Medicine, 389(12), 1069-1084.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.