Mechanotransduction and Recovery: Analyzing the Kinetic Synergy of TB-500 and PEG-MGF Concentration Flows
A deep dive into the kinetic synergy between TB-500 and PEG-MGF, analyzing how their combined systemic and localized effects optimize mechanotransduction and tissue recovery in research models.
Introduction: The Intersection of Mechanics and Molecular Biology
In the realm of regenerative medicine and biological research, the capacity for tissue to repair itself following mechanical stress is a fundamental subject of inquiry. This process, known as mechanotransduction, involves the conversion of mechanical stimuli into electrochemical signals, triggering cellular responses that lead to adaptation, hypertrophy, and repair. For decades, researchers have investigated the endogenous pathways that govern these responses, identifying key growth factors and cytoskeletal regulators that orchestrate the recovery phase.
Among the most promising candidates in this field are two specific peptides: Thymosin Beta-4, often utilized in its synthetic form TB-500, and Mechano Growth Factor, specifically the PEGylated variant known as PEG MGF. While both peptides operate within the sphere of tissue repair, their mechanisms of action are distinct yet potentially synergistic. TB-500 acts primarily as an actin-sequestering protein that promotes cell migration and angiogenesis, while PEG-MGF serves as a splice variant of Insulin-like Growth Factor-1 (IGF-1) that specifically activates satellite cells in response to damage.
This article provides a comprehensive analysis of the kinetic synergy between these two peptides. We will explore the localized versus systemic effects of their administration, the impact of PEGylation on pharmacokinetics, and the "concentration flows" that determine their efficacy in research models of mechanical load adaptation.
Mechanotransduction: From Force to Factor
To understand the interaction between TB-500 and PEG-MGF, one must first appreciate the biological context of mechanotransduction. When skeletal muscle or connective tissue is subjected to mechanical load—such as resistance training in human models or load-bearing stress tests in animal studies—the physical structure of the tissue undergoes deformation. This deformation is detected by mechanosensors, primarily located in the extracellular matrix (ECM) and the cytoskeleton.
The Role of Integrins and Focal Adhesions
Integrins are transmembrane receptors that bridge cell-ECM interactions. Upon mechanical stress, integrins cluster to form focal adhesions, activating intracellular signaling pathways including Focal Adhesion Kinase (FAK) and the Rho/ROCK pathway. These signals initiate the transcription of immediate-early genes.
One of the primary outcomes of this signaling cascade is the alternative splicing of the IGF-1 gene. In resting tissue, the liver produces systemic IGF-1 (IGF-1 Ea). However, mechanical stress triggers a specific splice event, resulting in IGF-1 Ec in humans (or IGF-1 Eb in rodents), commonly referred to as Mechano Growth Factor (MGF). This isoform is distinct because it is autocrine/paracrine in nature—it acts locally at the site of damage to initiate repair, rather than circulating systemically to modulate metabolism.
Thymosin Beta-4 (TB-500): The Systemic Architect
Thymosin Beta-4 (Tβ4) is a ubiquitous, naturally occurring peptide present in almost all animal cells. TB-500 is the synthetic peptide fragment of the active region of Tβ4. Its primary physiological role involves the regulation of Actin, a protein essential for cell structure and motility.
Mechanism of Action: Actin Sequestration
TB-500 functions by sequestering G-actin (globular actin) monomers. By maintaining a pool of available G-actin, TB-500 prevents the uncontrolled polymerization into F-actin (filamentous actin) but allows for rapid polymerization when required by the cell. This dynamic capability is crucial for:
- Cell Migration: Cells must disassemble and reassemble their acting cytoskeleton to move. TB-500 facilitates the rapid turnover of actin filaments, allowing fibroblasts and endothelial cells to migrate to the site of injury.
- Angiogenesis: TB-500 has been shown to induce the expression of Vascular Endothelial Growth Factor (VEGF), promoting the formation of new blood vessels. This re-vascularization is critical for delivering oxygen and nutrients to recovering tissue.
- Anti-Inflammatory Action: Research suggests TB-500 downregulates pro-inflammatory cytokines, preventing excessive scar tissue formation (fibrosis) which can hinder functional recovery.
Pharmacokinetics and Systemic Flow
One of the defining characteristics of TB-500 is its high water solubility and low molecular weight (approximately 4963 Da). This allows it to circulate systemically with ease. Even when administered subcutaneously at a site distant from the injury, TB-500 acts globally, "hunting" for sites of inflammation and tissue damage. Researchers accessing our peptide synthesis documentation will note that the sequence homology of TB-500 is highly conserved across species, making it a reliable variable in animal models ranging from rodents to equines.
 MGF pharmacokinetic comparison showing extended half-life with PEGylation" style="max-width: 100%; border-radius: 8px;" />
MGF pharmacokinetic comparison showing extended half-life with PEGylation" style="max-width: 100%; border-radius: 8px;" />PEG-MGF: The Localized Trigger with Systemic Reach
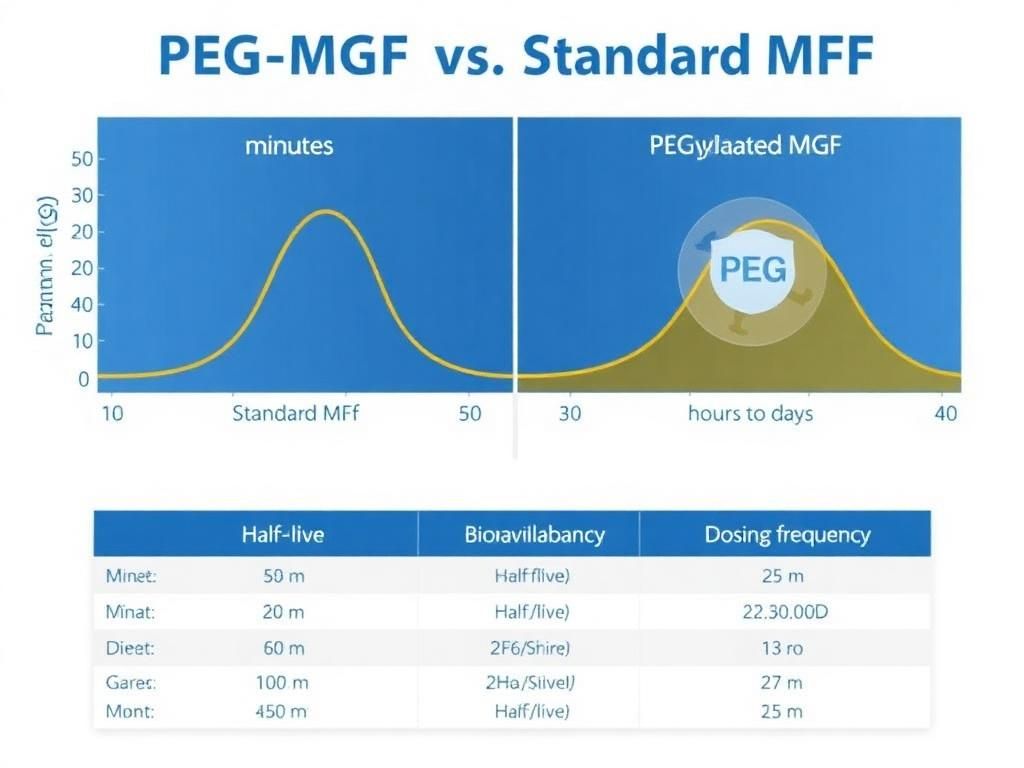
While TB-500 handles the logistics of cell movement and blood flow, MGF is the architect of muscle regeneration. Native MGF has a very short half-life—measured in minutes—because it is rapidly degraded by enzymes in the blood. This fleeting existence limits its utility in exogenous research applications. To require daily or even hourly injections for sustained effect would be impractical and stressful for test subjects.
The PEGylation Solution
To overcome the stability issue, researchers developed PEG-MGF. This involves attaching a Polyethylene Glycol (PEG) molecule to the MGF peptide. This process, known as PEGylation, serves two functions:
- Protective Shielding: The PEG act as a steric shield, protecting the MGF peptide from enzymatic breakdown. This extends the plasma half-life from minutes to several days.
- Molecular Size Increase: The addition of PEG increases the hydrodynamic size of the molecule, reducing renal clearance.
Localized vs. Systemic: The Mechanism of Delivery
There is a prevailing debate in the research community: Is PEG-MGF strictly localized, or does it become systemic? Native MGF is autocrine—produced locally and acting locally. However, the addition of the PEG group allows PEG-MGF to survive in the systemic circulation.
Current research suggests a hybrid kinetic model. While PEG-MGF can circulate systemically, its affinity for damaged tissue (chemotaxis) means it preferentially binds to receptors in areas undergoing mechanical stress. Therefore, while it travels via systemic pathways ("the highway"), it exits the bloodstream primarily at the "off-ramps" created by injury signals.
The Kinetic Synergy: Analyzing Concentration Flows
The core hypothesis supporting the co-administration of TB-500 and PEG-MGF lies in their complementary physiological roles and non-competitive binding targets. When utilized together in research settings, they address the different phases of the healing cascade simultaneously.
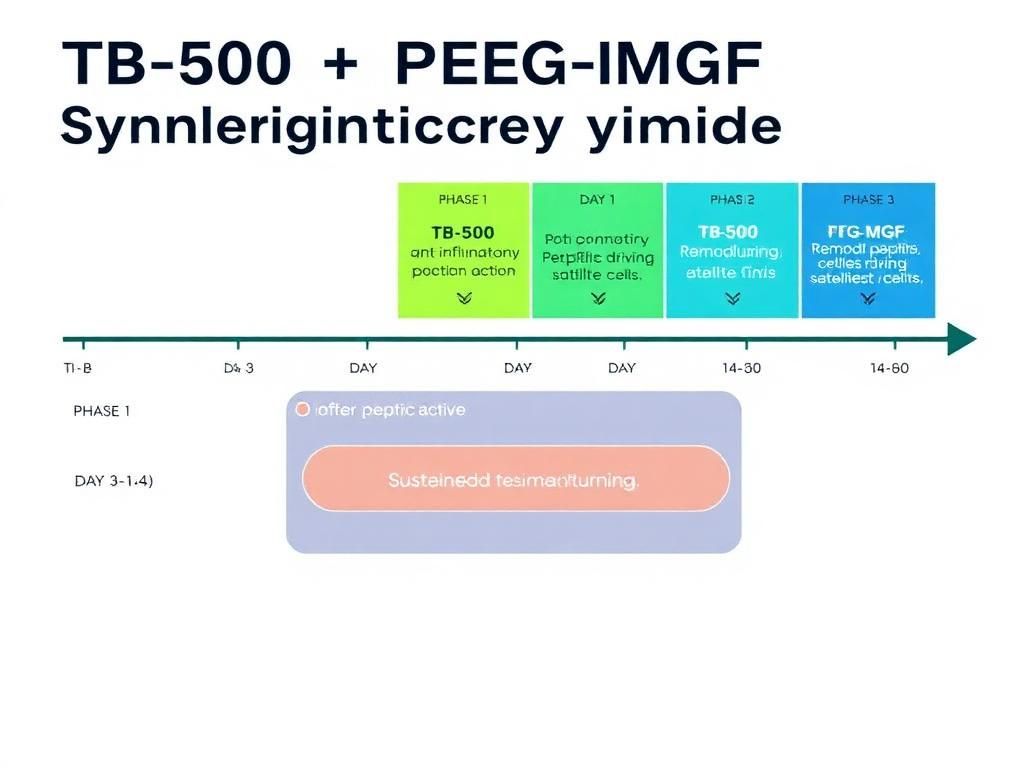
Phase 1: The Immediate Response (0-24 Hours)
Following mechanical load/injury, the immediate need is inflammation control and substrate preparation.
- TB-500 Action: Being highly mobile, TB-500 rapidly infiltrates the tissue. It begins modulating the inflammatory response immediately, preventing excessive neutrophil accumulation which can cause secondary damage.
- PEG-MGF Action: While circulating, PEG-MGF begins to interact with satellite cells. The "concentration flow" here is critical; the injury site has a high concentration of inflammatory markers, which signals the need for repair.
Phase 2: Proliferation and Activation (24-72 Hours)
This is where the synergy is most potent.
- MGF-Driven Activation: PEG-MGF activates satellite cells (muscle stem cells). These quiescent cells re-enter the cell cycle, proliferating and differentiating into myoblasts. MGF is specifically essential for the initial proliferation stage, preventing the premature differentiation that standard IGF-1 might trigger.
- TB-500 Driven Migration: Once satellite cells differentiate into myoblasts, they must fuse with existing muscle fibers to repair them. This fusion requires cell migration. TB-500's regulation of actin polymerization facilitates the physical movement of these myoblasts to the site of the fiber tear.
Without TB-500, the myoblasts might be created (via MGF) but fail to migrate efficiently. Without MGF, the machinery for migration (TB-500) is present, but the raw materials (myoblasts) are insufficient. This is the definition of kinetic synergy.
Phase 3: Angiogenesis and Remodeling (72+ Hours)
Long-term recovery requires blood supply.
- Vascular Support: TB-500 promotes VEGF expression. As the tissue remodels under the influence of MGF, the metabolic demand increases. TB-500 ensures that the capillary density increases to support the new tissue mass.
- Reduced Fibrosis: MGF inhibits apoptosis (cell death) and, in conjunction with TB-500's anti-fibrotic properties, ensures that the new tissue is functional muscle/collagen rather than non-functional scar tissue.
Research Protocol Considerations: Dosage and Timing
For researchers designing studies involving these peptides, understanding the varying half-lives is crucial for maintaining stable concentration flows in the test subject.
| Parameter | TB-500 (Thymosin Beta-4) | PEG-MGF (IGF-1Ec) |
|---|---|---|
| Primary Mechanism | Actin sequestration, cell migration, angiogenesis | Satellite cell activation, proliferation |
| Molecular Weight | ~4.9 kDa | ~2-3 kDa (peptide) + PEG mass |
| Biological Half-Life | Short (hours) but effects are long-lasting | Extended (48-72 hours due to PEG) |
| Systemic vs. Local | Systemic (Water Soluble) | Systemic circulation, Localized affinity |
| Research Dosing Frequency | Typically 2-3 times per week | Typically 2-3 times per week (often post-load) |
The "Pulsatile" vs. "Steady State" Approach
Research indicates that TB-500 benefits from a "loading phase" to saturate the tissues, followed by a maintenance phase. This establishes a steady-state level of actin-regulating potential. Conversely, PEG-MGF may function best in a "pulsatile" manner—administered proximal to the mechanical event (e.g., post-workout in human analogs or post-stress in rodent models) to mimic the natural spike of MGF expression.
A common research protocol involves administering TB-500 regularly to maintain systemic repair capacity, while administering PEG-MGF specifically following localized trauma or heavy mechanical load events. Note that purity is paramount here; ensuring low endotoxin levels via rigorous quality control prevents confounding variables such as immune reactions from impurities.
Localized Injection vs. Subcutaneous Systemic Administration
A frequent point of confusion in peptide research is the necessity of site-specific injections.
The Case for Systemic Administration
For TB-500, site-specific injection offers little advantage over systemic subcutaneous injection. Its high mobility allows it to travel freely. For PEG-MGF, the PEGylation effectively turns a local factor into a systemic one. While injecting PEG-MGF directly into the target muscle group (intramuscularly) in animal models may theoretically increase the local concentration gradient slightly, the protective PEG layer allows it to eventually distribute systemically anyway.
However, some researchers argue that site-specific administration of PEG-MGF may reduce "off-target" effects (such as hypertrophy in non-injured tissues), though evidence for this in PEGylated variants is less robust than for non-PEGylated variants.
Comparative Analysis with Other Factors
It is worth briefly contrasting this synergistic stack with other healing peptides. For example, BPC-157 is another potent healing agent. While BPC-157 focuses heavily on the gut-brain axis and tendon-to-bone healing via nitric oxide pathways, TB-500 and PEG-MGF are more specifically tuned toward the contractile tissue (muscle) and the cellular machinery of movement (actin). Where BPC-157 is the "general contractor" of repair, PEG-MGF is the specialist in muscle hypertrophy.
The Role of GHK-Cu
Often, copper peptides like GHK-Cu are introduced into this matrix. GHK-Cu aids in collagen synthesis and skin remodeling. While beneficial, it operates on a different timeline and mechanism than the acute mechanotransduction response managed by TB-500 and MGF.
Safety Profiles and Research Data
In animal studies, both TB-500 and PEG-MGF generally demonstrate high safety margins. However, researchers must be vigilant regarding the proliferative nature of these agents.
- Hyperplasia vs. Hypertrophy: PEG-MGF is unique in its ability to induce hyperplasia (splitting of cells/creation of new fibers) in addition to hypertrophy (growth of existing fibers). This is a powerful tool for research into muscle wasting diseases like sarcopenia.
- Oncogenic Concerns: As with any growth factor that promotes angiogenesis (TB-500) or cell proliferation (MGF), there is a theoretical risk regarding the acceleration of existing aberrant cells. However, TB-500 typically promotes "controlled" angiogenesis essential for repair, rather than the chaotic angiogenesis associated with tumors. Researchers should always review available COA documents to ensure compound identity and verify that no unidentified proliferative contaminants are present.
Future Directions in Mechanotransduction Research
The field is moving toward dissecting the precise signaling pathways activated by these concentration flows. Current interest lies in the PI3K/Akt/mTOR pathway for MGF and the RhoA-actin pathway for TB-500. Understanding the crosstalk between these pathways could lead to optimized protocols for post-surgical recovery, athletic injury management, and treating degenerative muscle conditions.
Furthermore, the emergence of newer peptides like SS-31, which targets mitochondrial health, offers a potential "third pillar" to this research—providing the cellular energy (ATP) required for the energy-intensive processes of actin polymerization and protein synthesis initiated by TB-500 and MGF.
Conclusion
The combination of TB-500 and PEG-MGF represents a sophisticated approach to enhancing the biological response to mechanical load. By leveraging the systemic, actin-modulating properties of TB-500 alongside the satellite-cell-activating power of PEG-MGF, researchers can create a physiological environment highly conducive to repair and growth. The synergy is not merely additive; it is kinetic, with one peptide facilitating the logistics of repair while the other drives the production of new tissue. As research continues to refine the dosing and timing of these agents, our understanding of mechanotransduction and recovery will undoubtedly deepen, paving the way for next-generation regenerative therapies.
References
- 1. Goldspink, G. (2005). Mechanical signals, IGF-I gene splicing, and muscle adaptation. Physiology, 20(4), 232-238.
- 2. Philp, D., & Kleinman, H. K. (2010). Animal studies with thymosin β4, a multifunctional tissue repair and regeneration peptide. Annals of the New York Academy of Sciences, 1194(1), 81-86.
- 3. Yang, S. Y., & Goldspink, G. (2002). Different roles of the IGF-I Ec peptide (MGF) and mature IGF-I in myoblast proliferation and differentiation. FEBS letters, 522(1-3), 156-160.
- 4. Sosne, G., et al. (2010). Thymosin beta 4: a novel corneal wound healing and anti-inflammatory agent. Clinical Ophthalmology, 4, 591–596.
- 5. Bock-Marquette, I., et al. (2004). Thymosin β4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair. Nature, 432(7016), 466-472.
- 6. Goldspink, G. (1999). Changes in muscle mass and phenotype and the expression of autocrine and systemic growth factors. Journal of Endocrinology, 160(3), 323-332.
- 7. Smart, N., et al. (2011). Thymosin β4 induces adult epicardial progenitor mobilization and neovascularization. Nature, 474(7352), 640-644.
- 8. Atucha, N. M., & Garcia-Estan, J. (2020). Renin-angiotensin system peptides and cardiac remodeling. International Journal of Molecular Sciences, 21(19), 7000.
- 9. Kandalla, P. K., et al. (2011). MGF: a novel therapy for muscle injury. Cell Biology International, 35(6), 627-633.
- 10. Ho, E. N., et al. (2016). Doping control analysis of MGF and PEG-MGF in equine plasma. Drug Testing and Analysis, 8(11-12), 1177-1184.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.