Kisspeptin-10: Reproductive Research
Explore Kisspeptin-10's role as the master regulator of the hypothalamic-pituitary-gonadal axis in fertility research.
Kisspeptin-10 represents one of the most significant discoveries in reproductive neuroendocrinology of the past two decades. This decapeptide, the biologically active fragment of the larger kisspeptin family, has emerged as the critical upstream regulator of gonadotropin-releasing hormone (GnRH) neurons, fundamentally reshaping our understanding of reproductive axis control. The discovery that kisspeptin signaling is essential for puberty and fertility has opened new frontiers in reproductive research.
Discovery and Scientific Significance
The significance of kisspeptin signaling in reproduction was established through landmark genetic studies published in 2003. Researchers led by Seminara and de Roux independently discovered that inactivating mutations in the kisspeptin receptor gene (KISS1R, formerly GPR54) cause idiopathic hypogonadotropic hypogonadism—a condition characterized by absent puberty and infertility due to GnRH deficiency. These findings positioned kisspeptin as an essential gatekeeper of reproductive function.
The term "kisspeptin" derives from the KISS1 gene, named after Hershey's Kisses chocolates by the discoverers at Penn State University in Hershey, Pennsylvania. Initially identified in 1996 as a metastasis suppressor in melanoma research, the reproductive significance of kisspeptin remained unknown until the pivotal 2003 discoveries linked its receptor to GnRH regulation.
Subsequent research has established Kisspeptin-10 as the minimal biologically active sequence capable of fully activating the KISS1R receptor. While longer forms including kisspeptin-54, kisspeptin-14, and kisspeptin-13 share the common C-terminal decapeptide sequence, Kisspeptin-10 retains complete receptor binding affinity and biological activity, making it the preferred form for research applications.
Molecular Structure and Receptor Pharmacology
Kisspeptin-10 consists of the amino acid sequence Tyr-Asn-Trp-Asn-Ser-Phe-Gly-Leu-Arg-Phe-amide, representing residues 112-121 of the precursor protein. The C-terminal amidation is essential for biological activity, as the free acid form shows dramatically reduced receptor binding. The aromatic residues, particularly the C-terminal phenylalanine-amide, are critical for receptor interaction and signal transduction.
The kisspeptin receptor (KISS1R) is a G protein-coupled receptor that primarily signals through Gαq/11 proteins, activating phospholipase C and increasing intracellular calcium and diacylglycerol levels. In GnRH neurons, this signaling cascade leads to neuronal depolarization, action potential firing, and GnRH secretion. The receptor demonstrates remarkable specificity, with kisspeptins being its only known endogenous ligands.
Research has characterized the structure-activity relationships of kisspeptin analogues extensively. Studies examining amino acid substitutions have identified positions critical for receptor binding versus those amenable to modification for improved pharmacokinetic properties. This work has informed the development of kisspeptin analogues with enhanced stability and prolonged duration of action for research applications.
Neuroanatomical Distribution and Function
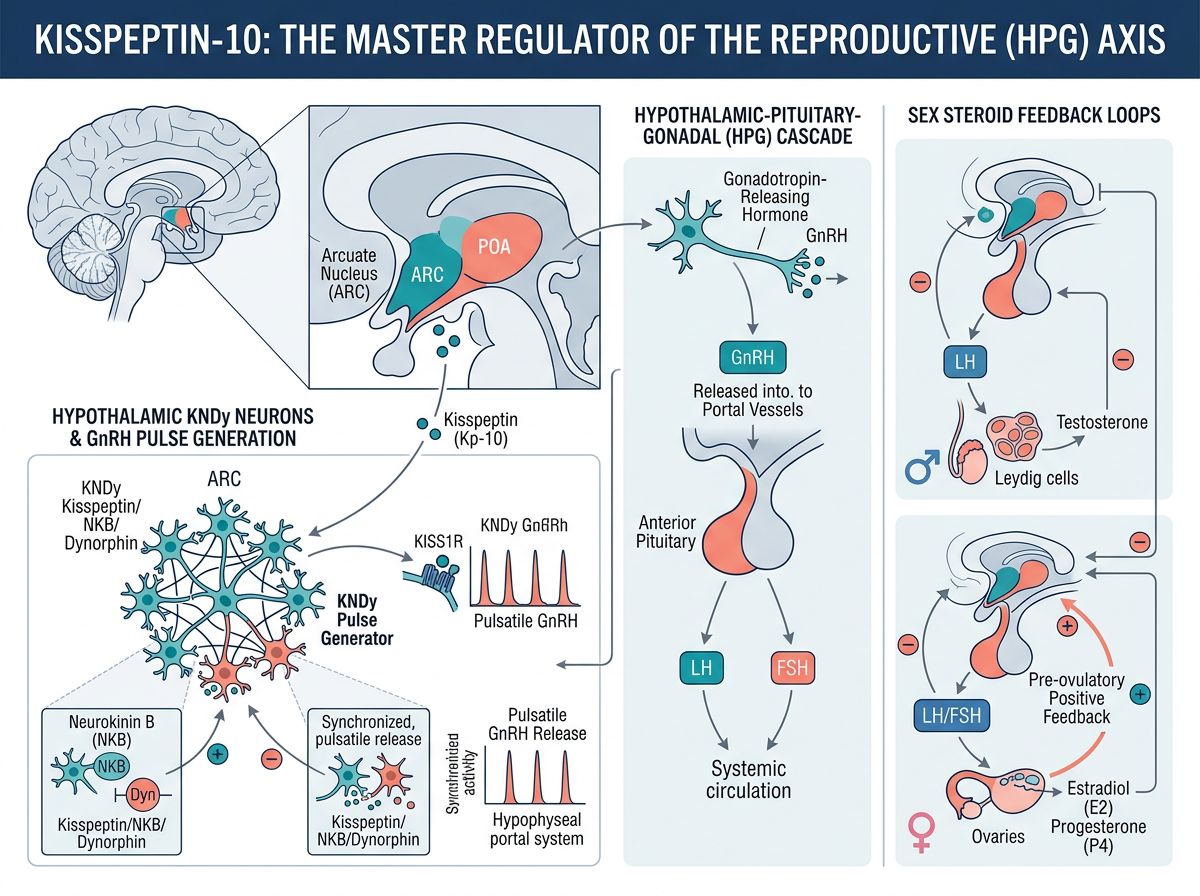
Kisspeptin-expressing neurons are found in two primary hypothalamic regions: the arcuate nucleus (ARC) and the anteroventral periventricular nucleus (AVPV) in rodents, with the preoptic area serving an analogous function in primates. These distinct neuronal populations serve different functions in reproductive regulation and respond differently to sex steroid feedback.
Arcuate nucleus kisspeptin neurons co-express neurokinin B (NKB) and dynorphin, earning the designation "KNDy neurons." These neurons are believed to generate the GnRH pulse generator rhythm through reciprocal interactions—NKB stimulates and dynorphin inhibits kisspeptin release, creating oscillatory patterns that drive pulsatile GnRH secretion essential for normal gonadotropin release.
AVPV/preoptic kisspeptin neurons respond positively to estrogen and are responsible for generating the preovulatory GnRH/LH surge in females. These neurons show pronounced sexual dimorphism, with females having substantially more kisspeptin neurons than males. This population is essential for the estrogen positive feedback mechanism that triggers ovulation.
Regulation of Puberty Onset
The role of kisspeptin in puberty initiation represents one of its most significant research applications. Studies have demonstrated that hypothalamic kisspeptin expression increases dramatically during pubertal development, temporally correlating with the awakening of the reproductive axis. This increased kisspeptin signaling drives the enhanced GnRH secretion characteristic of puberty.
Research examining the factors that trigger pubertal kisspeptin activation has identified metabolic signals as important regulators. Leptin, the adiposity hormone, appears to permit puberty onset by acting on kisspeptin neurons, potentially explaining the relationship between body composition and pubertal timing. Studies have shown that leptin receptor expression on kisspeptin neurons is necessary for normal pubertal development.
The concept of a "kisspeptin brake" on puberty has emerged from research demonstrating that inhibitory factors actively suppress kisspeptin expression during childhood. The MKRN3 gene, identified through studies of familial central precocious puberty, encodes a protein that inhibits kisspeptin neuron activity. Decreased MKRN3 expression during development may release this inhibition, contributing to puberty initiation.
GnRH Neuron Stimulation Mechanisms
Kisspeptin-10 administration produces robust, dose-dependent stimulation of GnRH release. Electrophysiological studies have demonstrated that kisspeptin directly depolarizes GnRH neurons, increasing their firing rate and synchronizing activity across the GnRH neuronal network. This stimulation occurs through KISS1R activation on GnRH neuron cell bodies and terminals.
Research has characterized the remarkable sensitivity of GnRH neurons to kisspeptin stimulation. Studies using electrophysiological recording have shown that kisspeptin can activate GnRH neurons at concentrations in the picomolar range, making it one of the most potent known stimulators of these neurons. This sensitivity underscores the physiological importance of kisspeptin signaling.
The temporal dynamics of kisspeptin-induced GnRH release have been extensively studied. Single kisspeptin administration produces a rapid increase in GnRH secretion, followed by a period of reduced responsiveness (desensitization) to subsequent stimulation. This desensitization phenomenon has important implications for research protocol design and potential therapeutic applications.
Sex Steroid Feedback Integration
Kisspeptin neurons serve as critical integrators of sex steroid feedback to the reproductive axis. Estrogen and testosterone regulate kisspeptin expression in a region-specific manner: negative feedback in the arcuate nucleus (decreasing kisspeptin expression) and positive feedback in the AVPV (increasing kisspeptin expression in response to rising estrogen levels).
Research has demonstrated that kisspeptin neurons express estrogen receptor alpha (ERα) and androgen receptors, enabling direct steroid hormone sensing. Studies using conditional knockout approaches have shown that ERα expression specifically in kisspeptin neurons is necessary for normal estrogen feedback regulation of the reproductive axis.
The integration of metabolic and stress signals with reproductive function also occurs at the level of kisspeptin neurons. Research has shown that conditions of energy deficit, chronic stress, and inflammatory states suppress kisspeptin expression, potentially explaining how these factors inhibit reproductive function. Understanding these regulatory mechanisms has implications for research into stress-related and metabolic reproductive disorders.
Research Applications in Reproductive Physiology
Kisspeptin-10 has become an essential research tool for investigating reproductive axis function. Administration studies in various species have demonstrated potent stimulation of LH and FSH release, providing a method to assess hypothalamic-pituitary responsiveness. This application is particularly valuable in research examining conditions affecting different levels of the reproductive axis.
Studies utilizing kisspeptin administration have helped differentiate hypothalamic from pituitary causes of hypogonadism. In subjects with hypothalamic dysfunction, kisspeptin can restore gonadotropin secretion, while pituitary disorders show absent responses. This diagnostic application has contributed to understanding the pathophysiology of various reproductive disorders.
Research examining pulsatile kisspeptin administration has explored its potential to restore physiological patterns of GnRH/gonadotropin release. Studies have demonstrated that repeated kisspeptin pulses can maintain LH pulsatility, potentially offering advantages over continuous administration which leads to receptor desensitization and decreased responsiveness.
Male Reproductive Research
In male subjects, Kisspeptin-10 potently stimulates testosterone production through increased LH secretion. Research has demonstrated dose-dependent increases in both LH and testosterone following kisspeptin administration, with effects observable within minutes of administration. This rapid hormonal response reflects the direct stimulation of GnRH neurons and subsequent pituitary activation.
Studies examining kisspeptin in male reproductive disorders have investigated its effects in conditions including hypogonadotropic hypogonadism, functional hypothalamic suppression, and aging-related testosterone decline. Research has shown that kisspeptin can restore testosterone production in conditions where hypothalamic GnRH release is impaired while the pituitary retains responsiveness.
The potential role of kisspeptin in spermatogenesis has also been explored. Beyond its effects on testosterone production, research has identified KISS1R expression in testicular tissue, suggesting possible direct effects on testicular function. Studies are investigating whether kisspeptin signaling influences Sertoli cell function or germ cell development independent of its effects on gonadotropin release.
Female Reproductive Research
Research in female subjects has examined kisspeptin's potential to trigger ovulation through stimulation of LH release. Studies have demonstrated that kisspeptin administration can induce LH surges of magnitude comparable to the natural preovulatory surge, suggesting potential applications in ovulation induction research. The ability to trigger ovulation through hypothalamic stimulation offers theoretical advantages over direct gonadotropin administration.
Investigations into kisspeptin for oocyte maturation triggering in assisted reproduction research have shown promising results. Studies have found that kisspeptin-triggered LH release can promote oocyte maturation while potentially reducing the risk of ovarian hyperstimulation syndrome compared to HCG triggering, likely due to the more physiological pattern of gonadotropin release.
Research examining kisspeptin in conditions of hypothalamic amenorrhea has demonstrated its ability to restore LH pulsatility in women with functional hypothalamic suppression. Studies in subjects with stress-related, exercise-induced, or eating disorder-associated amenorrhea have shown that kisspeptin can acutely stimulate gonadotropin release, indicating preserved pituitary and gonadal responsiveness in these conditions.
Metabolic Interactions Research
The interaction between kisspeptin signaling and metabolic status represents an active area of investigation. Research has demonstrated that kisspeptin neurons express receptors for metabolic hormones including leptin, insulin, and ghrelin, positioning them as integrators of nutritional information with reproductive function.
Studies examining kisspeptin in obesity and metabolic syndrome have revealed complex relationships between adiposity and reproductive axis function. While kisspeptin can stimulate gonadotropin release in obese subjects, the response magnitude may differ from lean individuals, suggesting that metabolic dysfunction affects hypothalamic kisspeptin signaling or downstream responsiveness.
Research into kisspeptin during fasting and caloric restriction has demonstrated suppression of hypothalamic kisspeptin expression, providing a mechanism for the reproductive suppression observed during energy deficit. Studies have shown that kisspeptin administration can partially overcome fasting-induced suppression of the reproductive axis, highlighting the role of decreased kisspeptin signaling in this response.
Behavioral and Psychological Research
Emerging research has explored effects of kisspeptin beyond reproductive hormone regulation. Studies using functional brain imaging have demonstrated that kisspeptin administration modulates activity in limbic brain regions involved in emotion processing and reward. These findings suggest potential effects on mood, motivation, and sexual behavior.
Research examining kisspeptin's effects on sexual arousal and desire has yielded intriguing findings. Studies have shown that kisspeptin can enhance brain responses to sexual stimuli and may improve measures of sexual aversion in some contexts. The relationship between reproductive hormone regulation and sexual behavior modulation represents a growing area of kisspeptin research.
Investigations into kisspeptin and emotional processing have demonstrated effects on responses to negative and positive emotional stimuli. Research using neuroimaging approaches has shown altered limbic activation following kisspeptin administration, suggesting potential roles in emotional regulation that extend beyond traditional reproductive endocrinology.
Pharmacokinetics and Research Considerations
Kisspeptin-10 has a relatively short plasma half-life of approximately 4-6 minutes due to rapid enzymatic degradation. This short half-life has important implications for research protocol design, necessitating either bolus injections for acute studies or continuous infusion for sustained stimulation. The rapid clearance also means that effects are quickly reversible upon discontinuation.
Research has explored various administration routes for kisspeptin, including intravenous, subcutaneous, and intranasal delivery. While intravenous administration provides the most reliable bioavailability, subcutaneous administration offers practical advantages for research protocols requiring repeated dosing. Intranasal delivery, though less studied, may provide a non-invasive alternative for future research applications.
The development of kisspeptin analogues with improved pharmacokinetic properties represents an active area of research. Modified kisspeptins with enhanced protease resistance and prolonged half-life have been synthesized and characterized. These analogues may offer advantages for research protocols requiring sustained kisspeptin receptor activation.
Future Research Directions
Research into kisspeptin signaling continues to expand across multiple domains. Studies examining the upstream regulators of kisspeptin neuron activity aim to understand how various physiological signals are integrated to control reproductive function. The identification of novel kisspeptin neuron afferents and their neurotransmitter systems remains an active area of investigation.
The potential development of kisspeptin-based approaches for reproductive applications continues to evolve. Research is exploring both agonist applications for conditions of reproductive insufficiency and antagonist approaches for applications requiring suppression of the reproductive axis. The unique position of kisspeptin as the final common pathway to GnRH neurons makes it an attractive target for reproductive research.
Investigations into the non-reproductive roles of kisspeptin signaling, including potential effects on metabolism, behavior, and cancer biology (relating to its original discovery as a metastasis suppressor), continue to reveal unexpected functions of this peptide system. These studies may identify novel research applications beyond traditional reproductive endocrinology.
Conclusion
Kisspeptin-10 has emerged as the master regulator of reproductive function, serving as the critical link between central nervous system inputs and GnRH neuron activation. From its role in puberty initiation to its integration of metabolic and stress signals with fertility, kisspeptin signaling represents a fundamental control mechanism in reproductive biology. The ongoing research into kisspeptin's diverse effects continues to reveal new aspects of this remarkable peptide system, with implications extending from basic reproductive physiology to potential novel research approaches for reproductive and metabolic conditions.
References
- 1. Seminara, S.B., et al. (2003). The GPR54 gene as a regulator of puberty. The New England Journal of Medicine, 349(17), 1614-1627.
- 2. de Roux, N., et al. (2003). Hypogonadotropic hypogonadism due to loss of function of the KiSS1-derived peptide receptor GPR54. Proceedings of the National Academy of Sciences, 100(19), 10972-10976.
- 3. Dhillo, W.S., et al. (2005). Kisspeptin-54 stimulates the hypothalamic-pituitary gonadal axis in human males. The Journal of Clinical Endocrinology & Metabolism, 90(12), 6609-6615.
- 4. Navarro, V.M., & Tena-Sempere, M. (2012). Neuroendocrine control by kisspeptins: role in metabolic regulation of fertility. Nature Reviews Endocrinology, 8(1), 40-53.
- 5. Comninos, A.N., et al. (2017). Kisspeptin modulates sexual and emotional brain processing in humans. The Journal of Clinical Investigation, 127(2), 709-719.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.