HMG: Gonadotropin Fertility Research
Explore HMG's dual FSH/LH activity, historical significance in reproductive medicine, and fertility research applications.
Human Menopausal Gonadotropin (HMG) represents one of the foundational compounds in reproductive medicine research. First extracted from the urine of postmenopausal women in the 1960s, this complex preparation contains both follicle-stimulating hormone (FSH) and luteinizing hormone (LH) activity, making it uniquely suited for research into gonadal function and reproductive physiology. The dual hormonal activity of HMG has established it as an essential tool in understanding the coordinated regulation of the reproductive axis.
Historical Development and Scientific Discovery
The story of HMG begins with the pioneering work of researchers in the early 1960s who recognized that postmenopausal women produce elevated levels of gonadotropins in their urine. This observation led to the development of extraction and purification techniques that enabled the isolation of bioactive gonadotropin preparations suitable for research and clinical applications.
The first successful use of HMG in inducing ovulation was reported by Lunenfeld in 1962, marking a watershed moment in reproductive medicine. This achievement demonstrated that exogenous gonadotropin administration could overcome certain forms of anovulatory infertility, opening new avenues for fertility research. The subsequent decades witnessed continuous refinement of extraction and purification methods, leading to preparations with improved consistency and reduced immunogenicity.
The historical significance of HMG extends beyond its direct applications to its role in advancing our understanding of reproductive endocrinology. Research utilizing HMG has contributed fundamental insights into folliculogenesis, steroidogenesis, and the hormonal regulation of reproductive function in both female and male subjects.
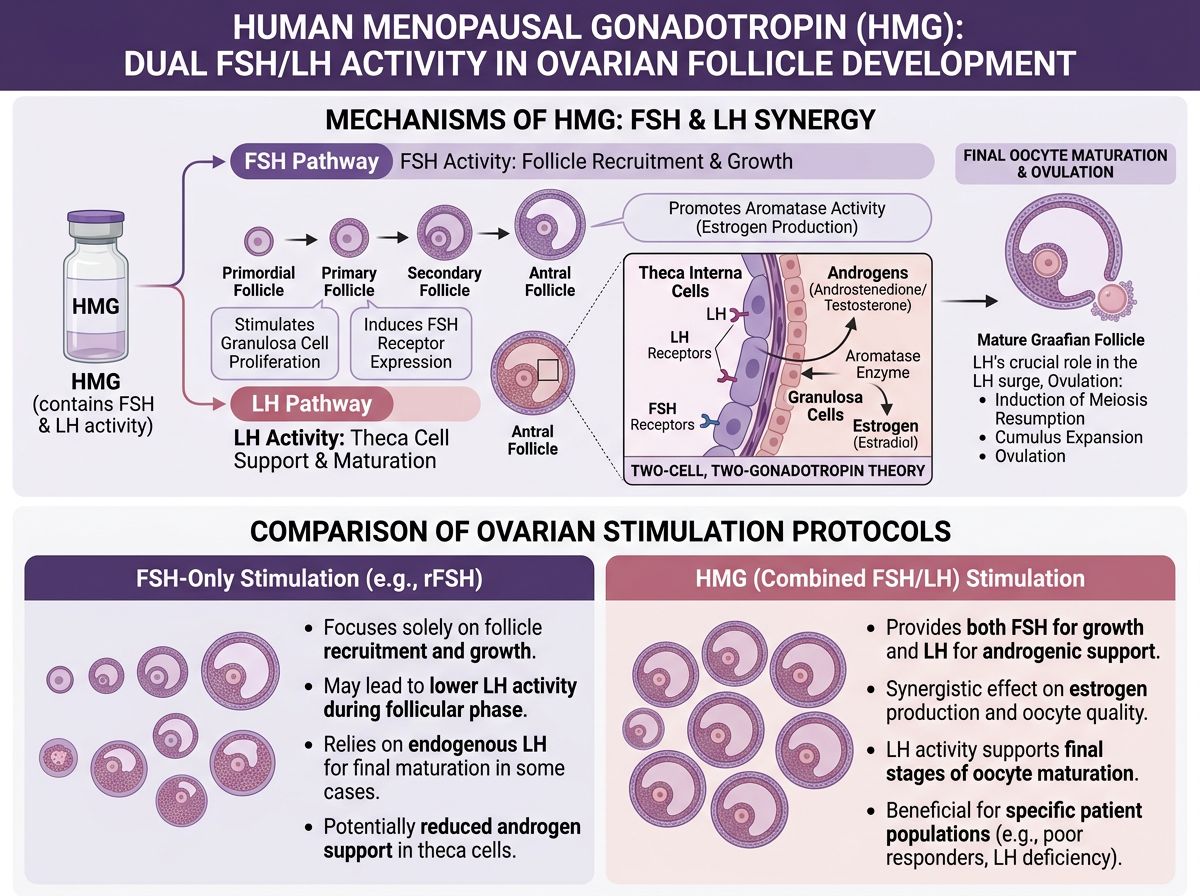
Composition and Biochemical Properties
HMG is characterized by its content of both FSH and LH activity, typically in an approximate 1:1 ratio, though this can vary between preparations. The FSH component is essential for follicular recruitment and growth, while the LH component supports theca cell function, androgen production, and final oocyte maturation. This combined activity distinguishes HMG from pure FSH preparations and provides a more physiologically complete gonadotropic stimulus.
The gonadotropins in HMG are glycoprotein hormones sharing a common α-subunit with other members of the glycoprotein hormone family, including HCG and thyroid-stimulating hormone. The unique β-subunits of FSH and LH confer receptor specificity and distinct biological activities. Extensive glycosylation of these hormones influences their receptor binding affinity, signal transduction potency, and metabolic clearance rates.
Early HMG preparations contained significant amounts of urinary proteins in addition to the gonadotropins, which could contribute to injection site reactions and immunogenic responses. Highly purified preparations, developed through advanced chromatographic techniques, contain substantially reduced non-gonadotropin protein content, improving tolerability while maintaining biological activity.
FSH Component: Folliculogenesis Research
The FSH activity within HMG primarily targets granulosa cells in the ovary, where it stimulates follicular growth and development. Research has extensively characterized the mechanisms through which FSH promotes folliculogenesis, including the induction of aromatase enzyme expression, which converts androgens to estrogens, and the upregulation of LH receptors on maturing granulosa cells.
Studies using HMG have contributed to understanding the concept of the FSH threshold—the minimum FSH concentration required to maintain follicular growth and prevent atresia. Research has demonstrated that follicles at different developmental stages exhibit varying FSH sensitivity, with more advanced follicles capable of continued growth at lower FSH concentrations.
The FSH component also influences cumulus cell function and oocyte quality. Research has shown that FSH signaling affects gap junction communication between the oocyte and surrounding cumulus cells, influences the production of hyaluronic acid-rich cumulus matrix, and modulates the expression of genes critical for oocyte competence and subsequent embryo development.
LH Component: Steroidogenesis and Ovulation
The LH activity in HMG serves multiple functions in reproductive research. In the ovary, LH stimulates theca cells to produce androgens, which serve as substrates for estrogen synthesis in adjacent granulosa cells. This two-cell, two-gonadotropin model of ovarian steroidogenesis represents a fundamental concept in reproductive endocrinology that has been elucidated in part through studies utilizing HMG.
Research has demonstrated that LH activity becomes increasingly important during late follicular development. As follicles mature, granulosa cells acquire LH receptors and become responsive to LH stimulation. This LH responsiveness is essential for the final stages of follicular maturation, including the resumption of meiosis and the acquisition of developmental competence by the oocyte.
The role of LH in HMG preparations has been a subject of considerable research interest. Studies have examined whether the LH component provides advantages over pure FSH preparations in certain contexts. Research suggests that LH activity may be particularly beneficial in hypogonadotropic hypogonadism, in older reproductive-age subjects, and in individuals with suboptimal responses to FSH-only stimulation.
Comparative Research: HMG vs. Pure FSH
A substantial body of research has compared the effects of HMG with recombinant FSH (rFSH) and other gonadotropin preparations. These comparative studies have examined endpoints including follicular development patterns, oocyte quality, fertilization rates, embryo development, and pregnancy outcomes in assisted reproductive technology research.
Meta-analyses examining the comparative effectiveness of HMG versus rFSH have yielded varying conclusions depending on the endpoints assessed and populations studied. Some analyses have suggested potential advantages for HMG in terms of live birth rates, while others have found comparable outcomes between preparation types. These discrepancies highlight the complexity of gonadotropin research and the influence of patient selection and protocol design on outcomes.
Research has also investigated the biological rationale for potential differences between HMG and pure FSH preparations. The presence of LH activity in HMG may provide benefits through enhanced androgen production supporting estrogen synthesis, improved oocyte maturation signaling, and modulation of follicular development dynamics. However, the optimal FSH:LH ratio remains an active area of investigation.
Male Reproductive Research Applications
While often discussed in the context of female reproduction, HMG has important applications in male reproductive research. In male subjects, FSH acts on Sertoli cells to support spermatogenesis, while LH stimulates Leydig cells to produce testosterone. The combined activity of HMG provides coordinated stimulation of both testicular cell populations.
Research examining HMG for male hypogonadotropic hypogonadism has demonstrated its ability to induce spermatogenesis in subjects with gonadotropin deficiency. Studies have characterized the time course of spermatogenic recovery during HMG treatment, typically requiring several months of therapy before sperm appear in the ejaculate, reflecting the approximately 74-day duration of human spermatogenesis.
Investigations into combination protocols utilizing HMG and HCG have explored optimal approaches for inducing spermatogenesis. Research suggests that pretreatment with HCG to establish adequate testosterone levels, followed by the addition of HMG or FSH to provide direct Sertoli cell stimulation, may represent an effective strategy in certain hypogonadotropic conditions.
Dosing Strategies and Protocol Research
Optimal dosing of HMG has been extensively studied across various research contexts. In ovarian stimulation protocols, starting doses typically range from 150-300 IU daily, with adjustments based on follicular response monitoring. Research has characterized factors influencing gonadotropin requirements, including age, body weight, ovarian reserve markers, and previous response history.
Studies examining step-up, step-down, and fixed-dose protocols have contributed to understanding of optimal stimulation strategies. Step-up protocols, involving gradual dose increases until follicular response is achieved, have been associated with reduced risk of hyperstimulation in some research contexts. Conversely, step-down protocols, starting with higher doses followed by reductions, may more closely mimic physiological FSH dynamics during the natural cycle.
Research into individualized dosing based on predictive markers represents a growing area of investigation. Studies have examined whether baseline characteristics including anti-Müllerian hormone (AMH) levels, antral follicle counts, and genetic polymorphisms can guide initial HMG dosing to optimize response while minimizing risks of over- or under-stimulation.
Ovarian Hyperstimulation Research
Ovarian hyperstimulation syndrome (OHSS) represents a significant concern in gonadotropin research, and studies utilizing HMG have contributed to understanding this condition's pathophysiology and prevention. OHSS is characterized by ovarian enlargement, increased vascular permeability, and fluid shifts that can range from mild to life-threatening in severity.
Research has identified risk factors for OHSS, including young age, low body weight, polycystic ovarian morphology, high estradiol levels, and large numbers of developing follicles. Studies have also characterized the role of vasoactive substances, particularly vascular endothelial growth factor (VEGF), in mediating the vascular changes characteristic of OHSS.
Investigations into prevention strategies have examined various approaches, including HCG trigger dose reduction, GnRH agonist triggering, dopamine agonist administration, and cycle cancellation criteria. These studies have contributed to development of evidence-based protocols aimed at maintaining research validity while minimizing hyperstimulation risk.
Quality Control and Standardization
The biological nature of urinary-derived HMG presents unique quality control challenges that have been subjects of ongoing research. Unlike recombinant preparations produced from defined cell lines under controlled conditions, urinary-derived products exhibit batch-to-batch variability influenced by donor characteristics and extraction processes.
Research has compared the biological activity of different HMG preparations using in vivo and in vitro bioassays. Studies have examined whether immunoassay-based potency measurements accurately predict biological activity, with some research suggesting discrepancies between immunological and functional assessments of gonadotropin content.
The development of highly purified HMG preparations has addressed some consistency concerns while maintaining the dual FSH/LH activity characteristic of the compound. Research comparing highly purified urinary preparations with recombinant gonadotropins continues to inform understanding of the relative advantages and limitations of each preparation type.
Emerging Research Directions
Contemporary research continues to explore novel applications and refinements in HMG utilization. Studies examining the role of LH activity in specific patient populations, optimal FSH:LH ratios for different clinical scenarios, and the biological significance of urinary versus recombinant gonadotropin sources represent active areas of investigation.
Research into the effects of gonadotropin glycoform composition on biological activity represents an emerging frontier. Studies suggest that variations in glycosylation patterns can influence receptor binding characteristics, signal transduction potency, and circulatory half-life. Understanding these relationships may enable development of gonadotropin preparations with optimized pharmacological properties.
Investigations into combination approaches utilizing HMG with other compounds, including growth hormone, DHEA supplementation, and various adjunctive therapies, continue to explore methods for optimizing reproductive research outcomes. These studies contribute to evidence-based protocol development and improved understanding of reproductive physiology.
Conclusion
Human Menopausal Gonadotropin maintains its position as an important tool in reproductive medicine research, offering unique combined FSH and LH activity that distinguishes it from pure gonadotropin preparations. From its historical origins to contemporary applications, HMG has contributed substantially to our understanding of reproductive endocrinology, follicular development, and the hormonal regulation of fertility. As research methodologies advance and our understanding of reproductive biology deepens, HMG continues to serve as a valuable compound for investigating the complex processes underlying reproduction in both female and male subjects.
References
- 1. Shoham, Z. (2002). The clinical therapeutic window for luteinizing hormone in controlled ovarian stimulation. Fertility and Sterility, 77(6), 1170-1177.
- 2. Lunenfeld, B. (2004). Historical perspectives in gonadotrophin therapy. Human Reproduction Update, 10(6), 453-467.
- 3. Coomarasamy, A., et al. (2008). Urinary hMG versus recombinant FSH for controlled ovarian hyperstimulation following an agonist long down-regulation protocol in IVF or ICSI treatment: a systematic review and meta-analysis. Human Reproduction, 23(2), 310-315.
- 4. Filicori, M., et al. (2002). Efficacy of low-dose human chorionic gonadotropin alone to complete controlled ovarian stimulation. Fertility and Sterility, 78(2), 343-349.
- 5. Balasch, J., & Fábregues, F. (2006). Is luteinizing hormone needed for optimal ovulation induction? Current Opinion in Obstetrics and Gynecology, 18(3), 265-272.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.