HCG: Gonadotropin Hormone Research
Explore HCG's molecular structure, physiological roles in pregnancy, and research applications in reproductive medicine.
Human Chorionic Gonadotropin (HCG) stands as one of the most clinically significant glycoprotein hormones in reproductive medicine. First identified over a century ago through its presence in the urine of pregnant women, this hormone has evolved from a simple pregnancy indicator to a multifaceted research compound with applications spanning fertility treatment, hormonal regulation, and metabolic research.
Molecular Structure and Biochemistry
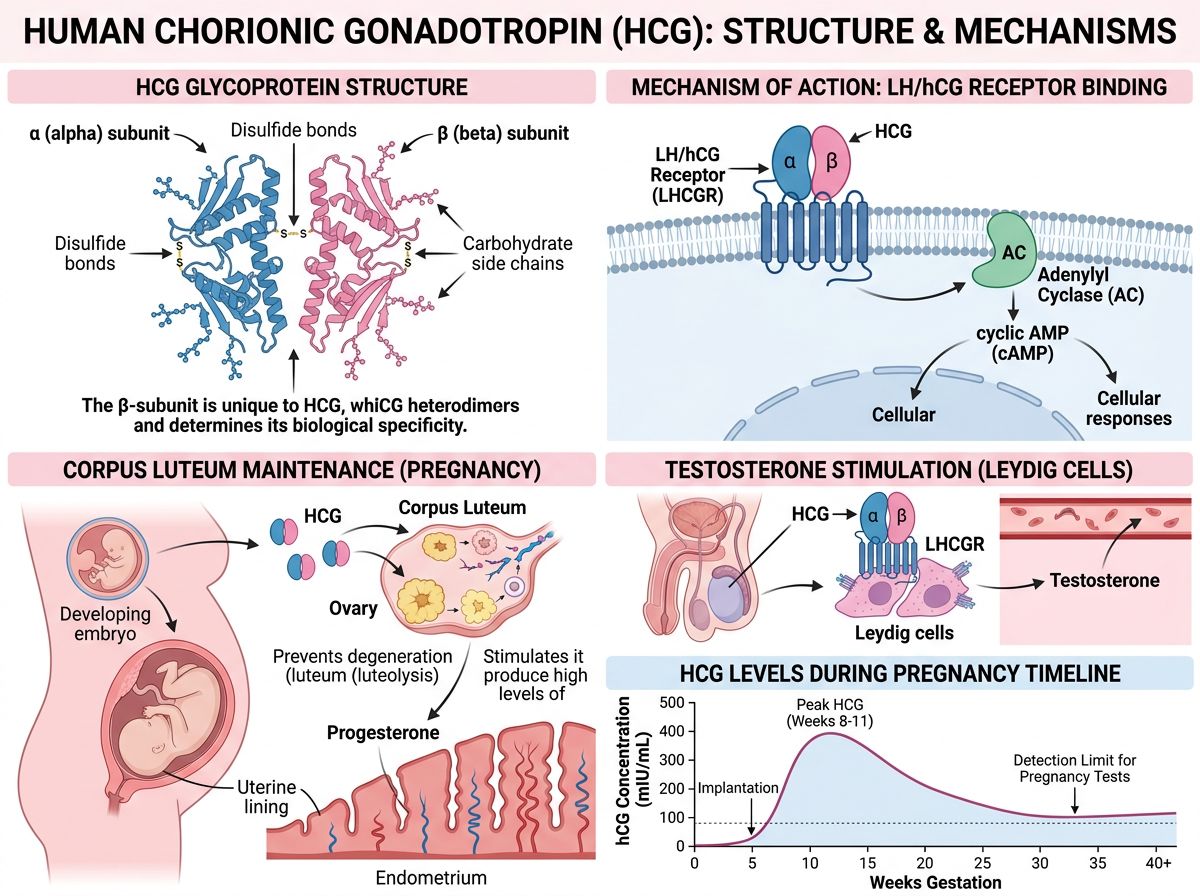
HCG is a heterodimeric glycoprotein composed of two non-covalently linked subunits: an alpha (α) subunit and a beta (β) subunit. The α-subunit, consisting of 92 amino acids, is shared with other glycoprotein hormones including luteinizing hormone (LH), follicle-stimulating hormone (FSH), and thyroid-stimulating hormone (TSH). This structural similarity is responsible for the overlapping biological activities observed among these hormones.
The β-subunit of HCG, comprising 145 amino acids, confers the hormone's unique biological specificity. Notably, the HCG β-subunit shares approximately 85% sequence homology with the LH β-subunit but contains an additional 24-amino acid carboxyl-terminal extension. This extension, along with extensive glycosylation patterns, accounts for HCG's significantly longer half-life compared to LH—approximately 24-36 hours versus 20 minutes.
The glycosylation of HCG plays crucial roles in its biological activity and metabolic stability. The hormone contains four N-linked and four O-linked oligosaccharide chains, with the extent and pattern of glycosylation varying depending on the source and physiological context. These carbohydrate moieties influence receptor binding affinity, signal transduction, and circulatory half-life.
Physiological Production and Regulation
Under normal physiological conditions, HCG is primarily produced by syncytiotrophoblast cells of the placenta during pregnancy. Production begins shortly after implantation, typically 6-12 days post-fertilization, and increases rapidly during the first trimester, reaching peak concentrations around weeks 8-11 of gestation. Subsequently, levels decline to a plateau that is maintained throughout the remainder of pregnancy.
The early surge in HCG production serves several critical functions during pregnancy. Most notably, HCG maintains the corpus luteum during early pregnancy, ensuring continued progesterone production until the placenta assumes this role around weeks 7-9 of gestation. This function is essential for maintaining the uterine lining and supporting embryo implantation and early development.
Beyond its placental production, low levels of HCG are produced by the pituitary gland in both men and non-pregnant women. Research has demonstrated pulsatile HCG secretion that follows the pattern of LH release, though at significantly lower concentrations. This pituitary HCG production increases substantially in postmenopausal women, coinciding with elevated gonadotropin levels.
Mechanism of Action and Receptor Biology
HCG exerts its biological effects primarily through binding to the luteinizing hormone/choriogonadotropin receptor (LHCGR), a G protein-coupled receptor expressed in various tissues. In the gonads, LHCGR is found on Leydig cells in the testes and theca and granulosa cells in the ovaries. Upon HCG binding, the receptor activates adenylyl cyclase, increasing intracellular cyclic AMP (cAMP) levels and triggering downstream signaling cascades.
The downstream effects of HCG-LHCGR activation are tissue-specific. In male subjects, receptor activation stimulates testosterone synthesis in Leydig cells through increased expression of steroidogenic enzymes. In female subjects, HCG promotes progesterone production, supports corpus luteum function, and facilitates oocyte maturation. These gonadal effects form the basis for many clinical and research applications of HCG.
Research has identified LHCGR expression in numerous extragonadal tissues, including the uterus, breast, prostate, brain, and adipose tissue. The physiological significance of extragonadal receptor expression remains an active area of investigation, with studies suggesting potential roles in immune modulation, angiogenesis, and tissue development.
Reproductive Medicine Applications
HCG has become an indispensable tool in reproductive medicine research and clinical practice. In assisted reproductive technologies, exogenous HCG administration serves as a trigger for final oocyte maturation prior to retrieval procedures. The hormone's LH-like activity induces resumption of meiosis in preovulatory follicles, mimicking the natural LH surge that precedes ovulation.
Studies examining the efficacy of HCG triggering in controlled ovarian stimulation have explored optimal dosing strategies and timing protocols. Research by Youssef et al. has compared various triggering agents and doses, contributing to evidence-based protocols that maximize oocyte yield while minimizing the risk of ovarian hyperstimulation syndrome (OHSS).
In male subjects, HCG research has focused on its ability to stimulate endogenous testosterone production. This application has garnered significant interest in studies examining hypogonadism treatment, fertility preservation, and hormonal optimization. Research protocols utilizing HCG often aim to maintain or restore testicular function while supporting physiological hormone levels.
Effects on Steroidogenesis
The steroidogenic effects of HCG have been extensively characterized in both in vitro and in vivo research. In testicular Leydig cells, HCG stimulation increases the expression of steroidogenic acute regulatory protein (StAR), which facilitates cholesterol transport into mitochondria—the rate-limiting step in steroid hormone synthesis. Subsequently, cholesterol is converted to pregnenolone and then through a series of enzymatic reactions to testosterone.
Research has demonstrated that HCG administration increases testosterone levels in a dose-dependent manner, with effects observable within hours of administration. Studies examining the kinetics of this response have characterized both acute and sustained effects on steroidogenesis, providing insights for dosing strategies in research protocols.
The effects of HCG on estrogen production have also been documented. Through aromatization of testosterone, HCG indirectly increases estradiol levels. Additionally, in ovarian tissue, HCG directly stimulates estrogen synthesis in granulosa cells. These estrogenic effects are relevant considerations in research protocol design and monitoring.
Spermatogenesis Research
The role of HCG in spermatogenesis has been investigated in numerous research contexts. While FSH is traditionally considered the primary driver of sperm production, testosterone produced in response to LH/HCG stimulation is essential for maintaining the seminiferous tubule environment and supporting spermatogenesis.
Research examining HCG as a therapeutic intervention for male infertility has yielded variable results, with outcomes depending on the underlying etiology. Studies in hypogonadotropic hypogonadism have demonstrated that combined FSH and HCG treatment can restore spermatogenesis in many subjects, highlighting the complementary roles of these hormones in male reproductive function.
Investigations into HCG monotherapy for maintaining spermatogenesis during testosterone replacement therapy have produced encouraging findings. Research suggests that low-dose HCG co-administration can preserve intratesticular testosterone concentrations and maintain sperm production in subjects receiving exogenous testosterone.
Weight Management Research
The potential role of HCG in weight management has been a subject of scientific investigation and debate. Early research by Simeons in the 1950s proposed that HCG could facilitate fat loss when combined with very-low-calorie diets. This hypothesis suggested that HCG might mobilize abnormal fat stores and reduce hunger during caloric restriction.
Subsequent controlled studies have produced mixed results regarding HCG's efficacy for weight loss. Meta-analyses of available research have concluded that evidence supporting HCG as a weight-loss agent is limited, and observed weight loss in studies is primarily attributable to severe caloric restriction rather than HCG-specific effects.
Despite inconclusive clinical evidence, mechanistic research continues to explore potential metabolic effects of HCG. Studies have examined the hormone's influence on adipose tissue lipolysis, metabolic rate, and appetite regulation. The identification of LHCGR in adipose tissue provides a theoretical basis for direct HCG effects on fat metabolism, though the physiological significance of this receptor expression remains to be fully elucidated.
Immunomodulatory Properties
Emerging research has revealed potential immunomodulatory properties of HCG. During pregnancy, the hormone may contribute to maternal immune tolerance of the developing embryo, preventing immunological rejection of the semi-allogeneic fetus. Studies have demonstrated that HCG can influence T-cell function, cytokine production, and regulatory immune cell populations.
Research examining HCG's effects on autoimmune conditions has explored whether the hormone's immunomodulatory properties might have therapeutic applications beyond pregnancy. Animal model studies have investigated HCG in the context of experimental autoimmune diseases, with some research suggesting potential protective effects.
The anti-inflammatory effects of HCG have also been documented in various research contexts. Studies have shown that HCG can suppress pro-inflammatory cytokine production and promote anti-inflammatory mediators. These findings have stimulated interest in HCG's potential role in conditions characterized by chronic inflammation.
Tumor Biology Research
The relationship between HCG and neoplastic disease has been extensively studied, given the hormone's ectopic production by various tumor types. Gestational trophoblastic diseases, including hydatidiform moles and choriocarcinoma, are characterized by abnormal HCG production and are routinely monitored using HCG measurements.
Beyond trophoblastic tumors, elevated HCG levels have been documented in testicular germ cell tumors, where the hormone serves as a valuable tumor marker for diagnosis, staging, and treatment monitoring. Research has characterized the prognostic significance of HCG levels and established guidelines for their interpretation in clinical management.
Interestingly, some research has explored potential anti-tumor properties of HCG in certain contexts. Studies have examined whether HCG or its variants might influence cancer cell growth, apoptosis, or metastasis. The dual nature of HCG in tumor biology—as both a tumor marker and potential therapeutic agent—continues to be an active area of investigation.
HCG Variants and Isoforms
Research has identified multiple HCG variants with distinct biological properties. Regular HCG produced by normal placental tissue differs from hyperglycosylated HCG, which predominates during early pregnancy and in certain tumors. These variants exhibit different receptor binding characteristics and signaling properties.
The free β-subunit of HCG represents another variant with potential biological significance. Unlike the intact heterodimer, free β-HCG has been associated with certain adverse pregnancy outcomes and tumor types. Research continues to characterize the distinct properties and clinical implications of various HCG isoforms.
Advanced analytical techniques have enabled detailed characterization of HCG glycoform patterns. Studies have shown that glycosylation profiles vary between pregnancy-derived HCG, tumor-produced HCG, and pituitary HCG. These differences may have implications for assay specificity and biological activity interpretation.
Research Considerations and Administration
In research settings, HCG is typically administered via intramuscular or subcutaneous injection. The hormone's relatively long half-life allows for less frequent dosing compared to LH, making it more practical for research protocols requiring sustained gonadotropic stimulation.
Dosing strategies in HCG research vary considerably depending on the application and desired endpoints. Fertility protocols often employ single high-dose administrations for ovulation triggering, while studies examining testosterone stimulation or testicular function maintenance may utilize lower doses administered over extended periods.
Recombinant HCG (r-hCG), produced through recombinant DNA technology, offers advantages over urinary-derived preparations, including higher purity, batch-to-batch consistency, and absence of urinary protein contaminants. Research comparing recombinant and urinary-derived preparations has generally demonstrated comparable efficacy, though subtle differences in glycosylation patterns exist.
Future Research Directions
The expanding understanding of HCG biology continues to open new research avenues. Investigations into extragonadal LHCGR expression and function may reveal novel physiological roles for HCG signaling beyond reproduction. Research examining HCG's potential neuroprotective, cardioprotective, and metabolic effects represents promising areas of exploration.
Advanced understanding of HCG variant biology may lead to the development of improved diagnostic assays and potentially to variant-specific therapeutic applications. Research into structure-activity relationships could enable the design of modified HCG analogues with enhanced selectivity or altered pharmacokinetic properties.
The integration of HCG research with advances in reproductive endocrinology, oncology, and metabolic medicine promises to yield new insights into this remarkable hormone's diverse biological functions. As research methodologies continue to evolve, our understanding of HCG's mechanisms and applications will undoubtedly expand, potentially revealing therapeutic opportunities beyond current applications.
Conclusion
Human Chorionic Gonadotropin represents a hormone of remarkable complexity and clinical significance. From its essential role in pregnancy maintenance to its applications in reproductive medicine research, HCG continues to be a subject of intensive investigation. The hormone's effects on steroidogenesis, potential immunomodulatory properties, and emerging roles in extragonadal tissues ensure that HCG research will remain a dynamic field for years to come. As our understanding of this multifaceted hormone deepens, new research applications and insights into fundamental reproductive biology are certain to emerge.
References
- 1. Cole, L.A. (2010). Biological functions of hCG and hCG-related molecules. Reproductive Biology and Endocrinology, 8, 102.
- 2. Fournier, T., et al. (2015). Human chorionic gonadotropin: Different glycoforms and biological activity depending on its source of production. Annales d'Endocrinologie, 77(2), 75-81.
- 3. Kero, J., et al. (2000). Thyroid stimulating hormone stimulates human chorionic gonadotropin secretion by the placenta. Journal of Clinical Endocrinology & Metabolism, 85(10), 3900-3906.
- 4. Choi, J., & Bhala, N. (2021). Human Chorionic Gonadotropin. StatPearls Publishing.
- 5. Youssef, M.A., et al. (2014). Human chorionic gonadotropin for triggering ovulation. Cochrane Database of Systematic Reviews.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.