Connective Tissue Sovereignty: Bioactive Modeling of the TB-500 and IGF-1LR3 Research Axis
Explore the synergistic research potential of TB-500 and IGF-1LR3 in connective tissue repair. This deep dive analyzes the intersection of actin cytoskeletal remodeling and the mTOR anabolic pathway for enhanced collagen synthesis and recovery.
Introduction: Defining the Connective Tissue Axis
In the realm of regenerative biology and peptide efficacy, the restoration of connective tissue integrity represents one of the most complex challenges. Connective tissues—comprising tendons, ligaments, the extracellular matrix (ECM), and fascial networks—possess notoriously low vascularity and slow metabolic turnover rates compared to skeletal muscle or epithelial tissue. This biological reality creates a bottleneck in healing processes, often leading to proliferation dead-ends or the formation of structurally inferior scar tissue (fibrosis).
To overcome these physiological limitations, researchers are increasingly turning toward multi-modal peptide signaling. The concept of "Connective Tissue Sovereignty" refers to the ability to pharmacologically direct the cellular environment toward functional restoration rather than fibrotic patch-repair. Central to this directive is the research axis of TB-500 (synthetic Thymosin Beta-4) and IGF-1LR3 (Long Arginine 3-IGF-1).
Separately, these peptides modulate distinct aspects of cellular machinery. TB-500 is fundamentally a G-actin sequestering peptide, essential for cytoskeletal mobility and angiogenesis. IGF-1LR3 is a potent mitogen, driving protein synthesis and cellular hyperplasia. When investigated in tandem, they present a hypothetical framework where the structural logistics of cell migration (TB-500) are met with the raw anabolic drive of cellular division (IGF-1LR3). This article explores the biochemical underpinnings of this axis, providing a comprehensive analysis for researchers investigating soft tissue repair models.
The Biological Substrate: Understanding the ECM Deficit
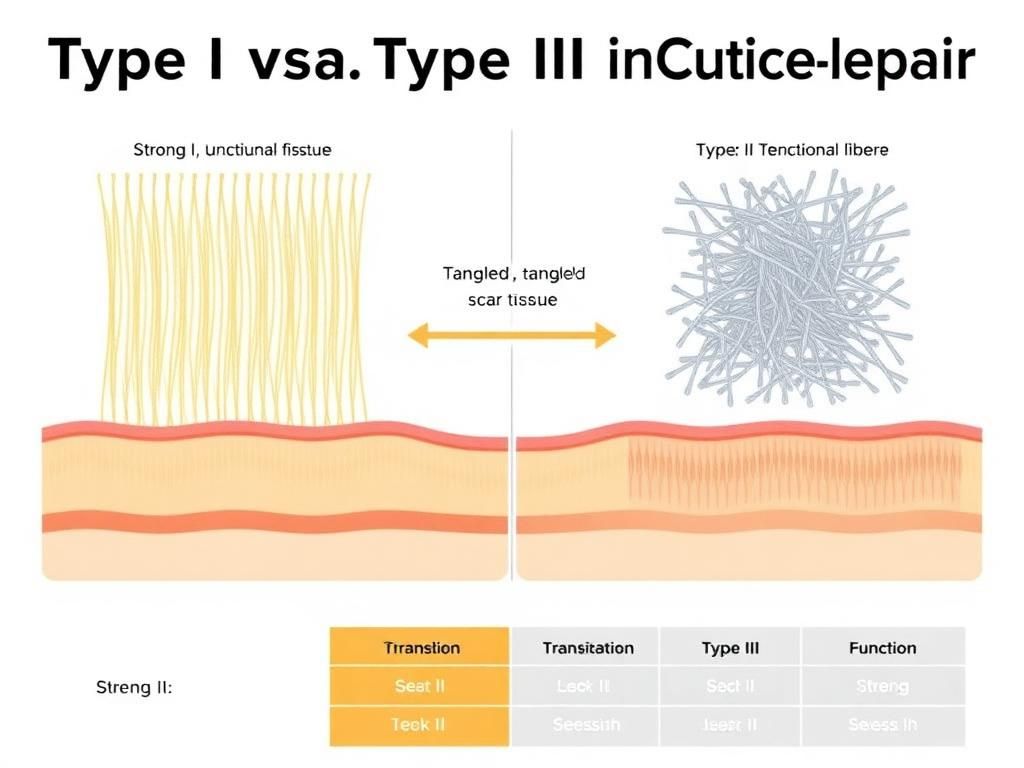
To understand why the TB-500 and IGF-1LR3 interaction is critical, one must first characterize the target environment: the Extracellular Matrix (ECM). The ECM is not merely scaffolding; it is a bioactive medium involved in signal transduction. In tendons and ligaments, the ECM is composed primarily of Type I collagen, proteoglycans, and elastin. Upon injury, the body’s default response is hemostasis followed by inflammation.
However, the subsequent proliferative phase is where the "SOvereignty" is often lost. Tenocytes (tendon cells) and fibroblasts often fail to migrate quickly enough to bridge the gap, or they produce Type III collagen—a disorganized, weaker variant—resulting in scar tissue. This is where the research into bioactive peptides becomes relevant. The goal is to modulate the ECM environment to favor Type I collagen synthesis, promote rapid vascularization (to bring nutrients), and enhance the migration speed of repair cells.
TB-500: The Cytoskeletal Modulator and Angiogenic Factor
Mechanism of Action: G-Actin Sequestration
TB-500 is a synthetic version of the naturally occurring 43-amino acid peptide Thymosin Beta-4 (Tβ4). Unlike growth factors that bind to surface receptors to initiate a kinase cascade, a primary mechanism of Tβ4 is intracellular. It binds to monomeric actin (G-actin) and sequesters it. This might seem counterintuitive at first—why inhibit actin polymerization? However, this sequestering creates a pool of available actin monomers that can be rapidly mobilized for filament polymerization when the cell needs to move.
Cell migration (chemotaxis) requires the rapid remodeling of the cytoskeleton. By maintaining a reservoir of G-actin, TB-500 allows tenocytes and fibroblasts to extend lamellipodia and move quickly toward the site of injury. Without this mobility, repair cells remain stagnant, and the wound gap is filled by random fibrin deposition rather than organized tissue.
Modulation of Inflammation and Angiogenesis
Beyond cytoskeletal value, TB-500 has demonstrated significant influence over the inflammatory cascade. Research suggests it downregulates nuclear factor-kappa B (NF-κB), a pivotal mediator of inflammatory responses. By tempering the inflammatory storm, TB-500 may prevent the excessive oxidative stress that leads to further tissue degradation during the early phases of injury.
Furthermore, Tβ4 is a known angiogenic driver. It stimulates the migration of endothelial cells, promoting the formation of new capillaries (neovascularization). For connective tissue, which is naturally hypovascular, this is crucial. The delivery of systemic nutrients, including amino acids and glucose, is rate-limited by blood flow. By enhancing capillary density, TB-500 fundamentally alters the metabolic potential of the injured site.
IGF-1LR3: The Anabolic Mitogen
The Modification: Why LR3?
Endogenous Insulin-like Growth Factor 1 (IGF-1) is a polypeptide hormone similar in molecular structure to insulin. It plays a key role in growth during childhood and has anabolic effects in adults. However, native IGF-1 has a very short half-life (less than 20 minutes) largely because it binds tightly to IGF Binding Proteins (IGFBPs), which render it biologically inactive.
IGF-1LR3 differs from native IGF-1 by the substitution of an arginine for glutamic acid at position 3 and an N-terminal extension of 13 amino acids. These structural changes drastically reduce its affinity for IGFBPs. Consequently, IGF-1LR3 remains biologically active in the system for 20 to 30 hours. This sustained bioavailability is what makes it a primary candidate for research into substantial tissue remodeling.
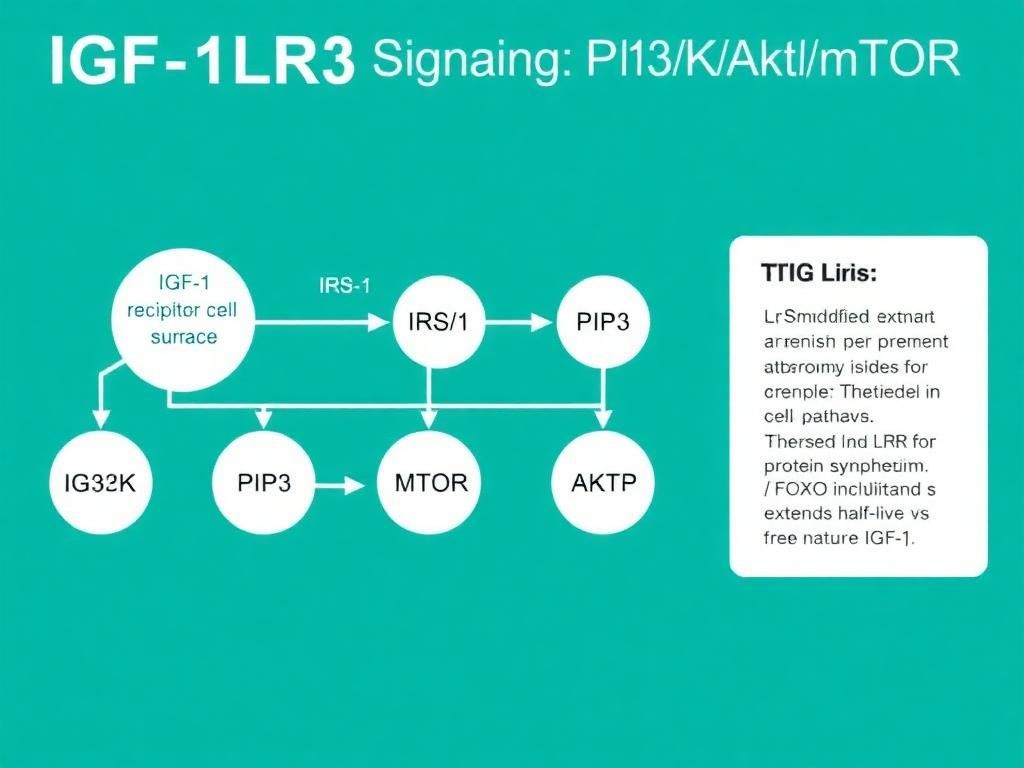
The mTOR Pathway and Protein Synthesis
The signaling sovereignty of IGF-1LR3 is exercised through the PI3K/Akt/mTOR pathway. Upon binding to the IGF-1 receptor (IGF-1R), a phosphorylation cascade initiates:
- IRS-1 Activation: Insulin Receptor Substrate 1 is phosphorylated.
- PI3K Recruitment: Phosphoinositide 3-kinase is recruited to the membrane.
- Akt Phosphorylation: Akt (Protein Kinase B) is activated, which inhibits catabolic pathways (like FoxO) and stimulates anabolic ones.
- mTORC1 Activation: The Mammalian Target of Rapamycin Complex 1 is the master regulator of protein synthesis. It activates S6K1 and inhibits 4E-BP1, leading to increased translation of mRNA into proteins.
In the context of connective tissue, this pathway drives the synthesis of collagen and proteoglycans. More importantly, IGF-1LR3 promotes hyperplasia—the splitting of cells to create new cells—rather than just hypertrophy (swelling of existing cells). For repairing a tear in a ligament, simply making existing cells bigger is insufficient; the tissue architecture requires more cells to bridge the gap.
The Intersection: Synergistic Mechanisms in Connective Tissue
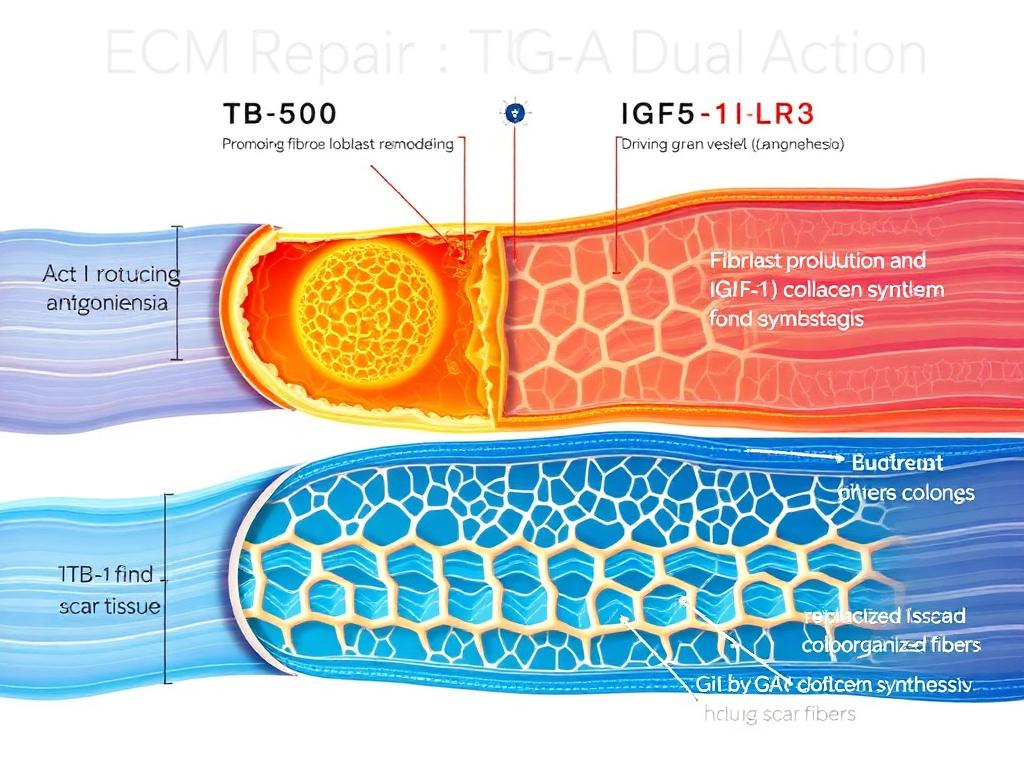
The "bioactive modeling" of these two agents implies that their combined effect is greater than the sum of their parts. When researching the TB-500 and IGF-1LR3 axis, we observe a complementary relationship between logistics and production.
1. The Supply Chain Synergy (Angiogenesis + Anabolism)
IGF-1LR3 drives a hyper-metabolic interaction. For a fibroblast to divide and synthesize collagen, it requires massive amounts of ATP, amino acids, and oxygen. However, as noted, scar tissue and damaged tendons typically have poor blood supply. If IGF-1LR3 is introduced to an environment with no supply chain, its efficacy is capped by nutrient availability.
TB-500 acts as the logistical engineer. By promoting angiogenesis (via VEGF expression and endothelial migration), TB-500 builds the "roads" (capillaries) that deliver the raw materials required by the IGF-1LR3-stimulated cells. This cross-talk ensures that the anabolic signal from IGF-1LR3 can be physically actualized by the tissue.
2. Migration vs. Proliferation
A specific challenge in resealing connective tissue tears is the "gap." Cells must physically move into the void of the injury to lay down new matrix. IGF-1LR3 stimulates the proliferation (division) of these cells, increasing the workforce. TB-500 stimulates the migration (motility) of this workforce.
Research suggests that without Tβ4-mediated actin remodeling, cells may divide but clump together efficiently rather than spreading across the injury matrix. The combination promotes a "divide and conquer" strategy: IGF-1LR3 creates the numbers, and TB-500 ensures they are distributed correctly across the ECM.
Comparative Analysis of Signaling Profiles
To differentiate the roles of these peptides in a research setting, the following table outlines their primary signaling vectors and physiological outcomes.
| Feature | TB-500 (Thymosin Beta-4) | IGF-1LR3 | The Axis (Combined) |
|---|---|---|---|
| Primary Mechanism | G-Actin Sequestering & Anti-inflammatory | IGF-1R Agonism & mTOR Activation | Structural Motility + Anabolic Drive |
| Cellular Effect | Migration, Survival, Angiogenesis | Proliferation (Hyperplasia), Differentiation | Accelerated Void Closure & Tissue Density |
| Half-Life | Short (subject but effects are lasting) | Long (~20-30 hours via IGFBP evasion) | Dual interaction window |
| Anti-Inflammatory | High (NF-κB downregulation) | Moderate (indirect recovery effects) | Comprehensive inflammatory modulation |
| Target Tissue | Systemic soft tissue, endothelials, cornea | Skeletal muscle, cartilage, bone, systemic | Refractory Tendinopathy, Muscle Tears, Ligaments |
Protocol Considerations in Research Settings
When modeling this axis in animal subjects or in vitro tissue cultures, researchers often explore staggered administration to align with the phases of healing. The healing process is generally divided into Inflammation, Proliferation, and Remodeling.
Phase 1: The Inflammatory/Acute Phase
In the immediate aftermath of tissue trauma, the priority is often the modulation of excessive inflammation and the initiation of cell migration. Research indicates that TB-500’s role is most critical here. Its ability to downregulate inflammatory cytokines protects the surviving tissue from oxidative damage. Early introduction of TB-500 establishes the pool of sequestered actin.
Phase 2: The Proliferative Phase
Once the acute inflammation subsides (typically days 3-7 in animal models), the metabolic demand shifts to protein synthesis. This is the optimal window for IGF-1LR3 activity. The cellular machinery is primed, and the foundational capillary networks initiated by TB-500 are forming. Introducing IGF-1LR3 here aims to maximize the deposition of collagen matrix.
Some researchers also investigate the inclusion of BPC-157 in this stack for its modulation of the NO (Nitric Oxide) pathway, though this adds another variable to the experiment. For pure connective tissue modeling, the TB-500/IGF-1LR3 axis is often studied for its specific impact on the actin-mTOR relationship.
Phase 3: Remodeling
During remodeling, the randomized collagen must be aligned. Mechanical loading (rehabilitation mimicry) is usually required here. Continued presence of TB-500 helps maintain tissue pliability, preventing the formation of rigid, restrictive adhesions. IGF-1LR3 administration is often tapered in this phase to prevent excessive tissue overgrowth, focusing instead on the maturation of the synthesized fibers.
Advanced Applications: Skeletal Muscle vs. Tendon
While this article focuses on connective tissue, the axis is highly relevant to skeletal muscle research. In muscle, satellite cells (stem cells) must be activated to fuse with existing muscle fibers. IGF-1LR3 is a potent activator of satellite cells. However, for the satellite cell to migrate to the site of damage on the myofibril, it requires the same cytoskeletal flexibility that TB-500 provides to bacteria cells.
Furthermore, muscle bellies are encased in fascia (connective tissue). A limiting factor in muscle growth is often the restriction of the fascial bag. By improving the elasticity and health of the fascia via TB-500, researchers hypothesize that IGF-1LR3 induced muscle hypertrophy can occur with less physical restriction, reducing the risk of compartment-syndrome-like dynamics or tearing at the myotendinous junction.
Integration with Other Peptides
Scientific inquiry rarely happens in a vacuum. The TB-500 and IGF-1LR3 axis is often compared to or combined with other agents.
- GHK-Cu: The copper peptide known for skin and collagen remodeling. Research combining BPC-157, TB-500, and GHK-Cu suggests a triad of healing: Nitric oxide modulation (BPC), Actin modulation (TB), and Collagen cross-linking (GHK-Cu).
- MGF (Mechano Growth Factor): MGF is a splice variant of IGF-1. While similar, MGF (specifically PEG MGF) is often viewed as better for the initial stem cell pool activation, while IGF-1LR3 is superior for the differentiation and protein synthesis phase. TB-500 remains the constant, facilitating the movement for both.
- Thymosin Alpha-1: While structurally related to Beta-4, Thymosin Alpha-1 is an immune modulator (T-cell maturation) and has less direct impact on actin and tissue repair. Care must be taken not to confuse the two in experimental ordering.
Quality Control and Experimental Integrity
The validity of any study regarding the TB-500/IGF-1LR3 axis hinges entirely on the purity of the substrates used. Peptide synthesis is a delicate process, particularly for longer chains like IGF-1LR3 (83 amino acids).
Purity and Synthesis Challenges
Impurities in peptide synthesis (such as truncation sequences or deletion sequences) can result in immunogenic responses that skew research data. For example, if an IGF-1LR3 batch contains high levels of incomplete chains, the receptor binding affinity may be unpredictable, or the half-life extension via the LR3 modification may be compromised.
At Alpha Carbon Labs, we emphasize peptide synthesis methods that prioritize specific folding and sequence accuracy. For TB-500, oxidation is a concern (methionine oxidation), which renders the peptide less effective in actin binding. Lyophilization techniques must be precise to ensure stability.
Verification
Researchers should always rely on High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) analysis. COA documents (Certificates of Analysis) provide the necessary transparency regarding purity (generally >98% is the research standard) and net peptide content. Without strict adherence to quality control, results attributed to the "axis" may actually be artifacts of contamination or degradation.
Future Directions in Bioactive Modeling
The future of connective tissue sovereignty lies in the precision of signaling. Current research is investigating the use of hydrogels infused with TB-500 and IGF-1LR3 for sustained, localized release in cartilage defects. This "depot" method could theoretically maintain high local concentrations while minimizing systemic exposure.
Additionally, genetic models are looking at the upregulation of endogenous Tβ4 in conjunction with exogenous IGF-1LR3 administration to treat chronic degenerative conditions like degradative tendinopathy or rotator cuff decay. The hypothesis is that by restoring the embryonic levels of Tβ4 (which are naturally high during fetal development where scarless healing occurs), combined with the potent adult growth factor IGF-1LR3, science can replicate a "regeneration" rather than "repair" phenotype in adult tissue.
Conclusion
The intersection of TB-500 and IGF-1LR3 represents a sophisticated approach to biological problem solving. By viewing connective tissue repair not as a singular event but as a coordinated sequence of migration, inflammation modulation, and anabolic synthesis, researchers can better utilize these peptides to influence healing outcomes.
TB-500 provides the "Sovereignty" over cellular movement and vascular access, while IGF-1LR3 provides the raw mitogenic signal required to rebuild tissue mass. Together, they form a research axis that addresses the fundamental deficits of the extracellular matrix: poor blood flow, slow cell turnover, and disorganized collagen deposition. As peptide synthesis technology advances, the ability to fine-tune this axis will likely yield significant breakthroughs in the treatment of musculoskeletal injuries.
References
- 1. Goldstein, A. L., et al. (2005). Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine, 11(9), 421-429.
- 2. Clemmons, D. R. (2012). Role of IGF-I in skeletal muscle mass maintenance. Trends in Endocrinology & Metabolism, 20(7), 349-356.
- 3. Sosne, G., et al. (2010). Thymosin beta 4 suppression of corneal NF-kappaB: a basis for anti-inflammatory efficacy. Experimental Eye Research, 90(3), 374-379.
- 4. Barton, E. R. (2006). Viral expression of insulin-like growth factor-I isoforms promotes different responses in skeletal muscle. Journal of Applied Physiology, 100(6), 1778-1784.
- 5. Philp, D., et al. (2004). Thymosin beta4 promotes angiogenesis, wound healing, and hair follicle development. Mechanisms of Ageing and Development, 125(2), 113-115.
- 6. Velloso, C. P. (2008). Regulation of muscle mass by growth hormone and IGF-I. British Journal of Pharmacology, 154(3), 557-568.
- 7. Smart, N., et al. (2011). Tymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature, 474, 640–644.
- 8. Schiaffino, S., & Mammucari, C. (2011). Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skeletal Muscle, 1(1), 4.
- 9. Ghannam, A., et al. (2013). High-Throughput Screening for Identification of Thymosin Beta 4 Inhibitors. Journal of Biomolecular Screening, 18(9), 1072–1080.
- 10. Tomas, F. M., et al. (1991). Insulin-like growth factor-I (IGF-I) and especially IGF-I variants are anabolic in dexamethasone-treated rats. Biochemical Journal, 282, 91-97.
- 11. Huff, T., et al. (2001). β-Thymosins, small acidic peptides with multiple functions. The International Journal of Biochemistry & Cell Biology, 33(3), 205-220.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.