ACE-031: Muscle Growth Research
Explore ACE-031's mechanism as a myostatin inhibitor and ActRIIB decoy receptor for muscle mass research.
Introduction to Myostatin and Muscle Regulation
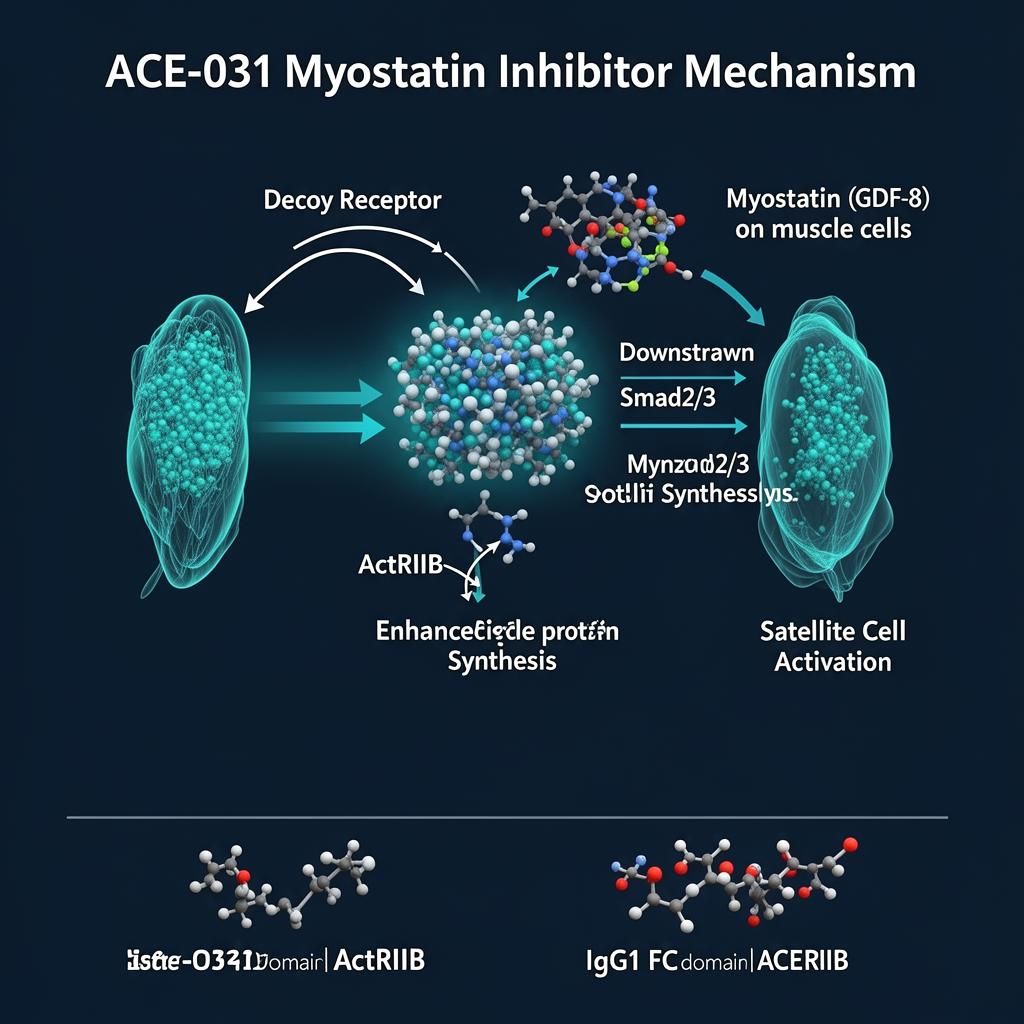
ACE-031 represents a groundbreaking approach to muscle regulation research, functioning as a soluble form of the activin receptor type IIB (ActRIIB). This fusion protein consists of the extracellular domain of human ActRIIB linked to the Fc portion of human IgG1, creating a "decoy receptor" that intercepts and neutralizes myostatin and related ligands before they can bind to their cellular receptors.
Understanding ACE-031 requires appreciation of myostatin's fundamental role in muscle biology. Myostatin, also known as growth differentiation factor-8 (GDF-8), is a member of the transforming growth factor-beta (TGF-β) superfamily that acts as a potent negative regulator of skeletal muscle mass. Discovered in 1997 by Se-Jin Lee and colleagues at Johns Hopkins University, myostatin provided the molecular explanation for the remarkable muscular phenotype observed in certain cattle breeds such as Belgian Blue and Piedmontese, which carry natural myostatin mutations.
The Biology of Myostatin Signaling
Myostatin is predominantly expressed in skeletal muscle tissue, where it is synthesized as an inactive precursor protein that undergoes proteolytic processing to release the mature, active form. This active myostatin circulates in blood and acts in both autocrine and paracrine fashion to regulate muscle growth. The signaling cascade begins when myostatin binds to ActRIIB on muscle cell surfaces, triggering receptor complex formation with type I receptors (primarily ALK4 and ALK5).
This receptor activation initiates Smad-dependent signaling pathways. Specifically, myostatin binding leads to phosphorylation of Smad2 and Smad3, which then complex with Smad4 and translocate to the nucleus. There, this transcriptional complex regulates the expression of genes involved in muscle growth, including downregulation of myogenic factors like MyoD and myogenin, and modulation of genes controlling protein synthesis and degradation.
The downstream effects of myostatin signaling include inhibition of satellite cell proliferation and differentiation (limiting muscle regenerative capacity), suppression of protein synthesis pathways (particularly the Akt/mTOR pathway), and activation of protein degradation through the ubiquitin-proteasome system. Together, these effects establish myostatin as a powerful brake on muscle growth and maintenance.
ACE-031 as a Myostatin Trap
ACE-031 functions by acting as a soluble ligand trap, sequestering myostatin and related TGF-β family members in the circulation before they can engage cell-surface receptors. The ActRIIB extracellular domain in ACE-031 retains high affinity for myostatin, while the Fc portion provides extended serum half-life and facilitates purification during manufacturing.
Importantly, ActRIIB is not exclusively a myostatin receptor—it also binds other TGF-β family ligands including activins, GDF-11, and bone morphogenetic proteins (BMPs). This broader binding profile means that ACE-031 inhibits multiple related signaling pathways, which may contribute to both enhanced muscle effects and potential side effects observed in research settings.
The structural design of ACE-031 as an Fc fusion protein offers several advantages for research applications. The Fc domain extends the molecule's circulating half-life from hours to weeks, allowing less frequent dosing. The bivalent nature of the Fc-fusion may also enhance ligand binding through avidity effects. Additionally, the well-characterized immunoglobulin framework provides favorable pharmacokinetic properties.
Preclinical Research Findings
Preclinical studies with ACE-031 and related ActRIIB inhibitors have demonstrated robust increases in muscle mass across multiple animal models. Research published in the Proceedings of the National Academy of Sciences demonstrated that ActRIIB-Fc fusion proteins produced significant increases in skeletal muscle mass in mice, with effects evident within weeks of treatment initiation.
Notably, these muscle mass increases occurred through hypertrophy of existing muscle fibers rather than hyperplasia (formation of new fibers), with observed increases in both type I (slow-twitch, oxidative) and type II (fast-twitch, glycolytic) muscle fiber cross-sectional areas. The muscle gains were accompanied by corresponding increases in strength and functional capacity in various muscle performance assays.
Animal studies also explored ACE-031's effects in disease models. In mdx mice—the murine model of Duchenne muscular dystrophy—ActRIIB inhibition produced meaningful increases in muscle mass and function despite the ongoing dystrophic process. These findings suggested potential therapeutic benefit in conditions where muscle wasting contributes to disease morbidity, generating considerable interest in clinical translation.
Research has also examined effects on other tissues. ActRIIB inhibition affects bone metabolism, with studies showing increased bone mineral density and altered bone formation markers. Effects on fat mass have also been observed, with some studies reporting reductions in adipose tissue alongside muscle gains—consistent with beneficial metabolic remodeling.
Clinical Research and Development
Based on promising preclinical results, ACE-031 advanced into clinical trials, initially targeting Duchenne muscular dystrophy (DMD)—a devastating X-linked genetic disorder caused by mutations in the dystrophin gene, leading to progressive muscle weakness and premature death. In DMD, loss of functional dystrophin renders muscle fibers susceptible to contraction-induced injury, leading to cycles of degeneration and incomplete regeneration that eventually exhaust the muscle's regenerative capacity.
Early-phase clinical studies demonstrated that ACE-031 was well-tolerated at various dose levels and produced measurable increases in lean body mass in healthy adult volunteers. These findings supported advancement into patient populations, where the potential benefit of increased muscle mass could be more meaningfully assessed.
However, clinical development encountered challenges related to the broad binding profile of ActRIIB. Because the receptor binds multiple ligands beyond myostatin, ACE-031 treatment was associated with unexpected effects, including minor nosebleeds and gum bleeding (epistaxis and gingival bleeding), as well as telangiectasias (dilated superficial blood vessels) in some participants. These findings were attributed to effects on other ActRIIB ligands involved in vascular biology, particularly BMP9 and BMP10, which are important for vascular endothelial function.
While these effects were generally mild and reversible, they prompted careful evaluation of the risk-benefit profile. The clinical program for ACE-031 was ultimately discontinued, though the scientific insights gained informed subsequent development of more selective myostatin inhibitors with narrower binding profiles designed to avoid off-target vascular effects.
Myostatin Pathway in Muscle Wasting Conditions
Research with ACE-031 and related compounds has significantly advanced understanding of how the myostatin pathway contributes to muscle wasting in various disease states. Elevated myostatin expression or signaling has been implicated in numerous conditions associated with muscle loss, including:
Muscular Dystrophies: In DMD and other muscular dystrophies, myostatin pathway inhibition may help preserve muscle mass despite ongoing degeneration. The muscle-building effects could complement strategies aimed at addressing the underlying genetic defect, such as gene therapy or exon skipping.
Age-Related Sarcopenia: Aging is associated with gradual loss of muscle mass and function (sarcopenia), contributing to frailty, falls, and loss of independence. Research suggests that myostatin levels may increase with age, potentially contributing to sarcopenic muscle loss. ActRIIB inhibition has shown promise in preclinical aging models.
Cancer Cachexia: Many cancers are associated with severe muscle wasting (cachexia) that contributes significantly to morbidity and mortality. Tumor-derived factors can activate muscle protein degradation pathways, and myostatin signaling may contribute to this process. Preclinical studies suggest that ActRIIB inhibition may help preserve muscle mass in cancer cachexia models.
Disuse and Immobilization: Muscle unloading during bed rest, immobilization, or spaceflight leads to rapid muscle atrophy. Research has explored whether myostatin inhibition could help preserve muscle mass during periods of disuse, with potential applications in rehabilitation and spaceflight medicine.
Comparison with Other Myostatin Inhibitors
ACE-031 represents one approach within a broader landscape of myostatin pathway inhibition strategies. Understanding these different approaches provides context for ACE-031's unique characteristics:
Myostatin-Specific Antibodies: Monoclonal antibodies that specifically bind and neutralize myostatin (such as stamulumab and domagrozumab) offer high selectivity but target only myostatin itself, not related ligands that also signal through ActRIIB. These may have narrower effects but potentially also more limited efficacy.
Propeptide and Follistatin Approaches: Myostatin's propeptide domain, which normally keeps myostatin inactive, and follistatin, a natural myostatin inhibitor, represent endogenous mechanisms of pathway regulation. Gene therapy approaches delivering follistatin have been explored in clinical trials.
Receptor-Specific Inhibitors: More selective ActRIIB ligand traps that preferentially bind myostatin and activins while sparing BMPs involved in vascular function have been developed in response to the clinical findings with ACE-031.
ACE-031's broader binding profile means it inhibits multiple negative regulators of muscle growth simultaneously, potentially producing more robust muscle effects. However, this same broad profile underlies the vascular effects that complicated clinical development, illustrating the trade-offs inherent in drug design.
Satellite Cells and Muscle Regeneration
A critical aspect of ACE-031's mechanism involves its effects on satellite cells—the muscle-resident stem cells responsible for regeneration and repair. Under normal conditions, satellite cells remain quiescent beneath the basal lamina of muscle fibers. In response to injury or growth signals, satellite cells activate, proliferate, and differentiate into myoblasts that either fuse with existing fibers (causing hypertrophy) or form new fibers (myogenesis).
Myostatin acts as a brake on satellite cell activity, maintaining quiescence and limiting proliferative capacity. By intercepting myostatin, ACE-031 releases this brake, enabling enhanced satellite cell activation and contribution to muscle growth. In muscular dystrophies, where the satellite cell pool is progressively depleted through repeated cycles of degeneration and regeneration, preserving satellite cell function is particularly important.
Research has demonstrated that myostatin inhibition can expand the satellite cell pool and enhance regenerative capacity following muscle injury. These effects may be especially relevant in conditions where ongoing muscle damage necessitates continuous regeneration, as in muscular dystrophies.
Effects on Muscle Fiber Type Composition
Skeletal muscle contains different fiber types with distinct metabolic and contractile properties. Type I (slow-twitch) fibers are oxidative and fatigue-resistant, while Type II (fast-twitch) fibers generate more force but fatigue more quickly. The balance between fiber types affects overall muscle function and metabolic health.
Research with ACE-031 and related ActRIIB inhibitors has examined effects on fiber type composition. While both fiber types undergo hypertrophy with myostatin inhibition, studies have reported preferential effects on Type II fibers in some contexts. This fiber-type specificity has implications for functional outcomes, as Type II fiber atrophy is particularly prominent in aging and certain disease states.
Understanding how ACE-031 affects fiber type distribution and metabolic properties helps predict functional outcomes and potential applications. For conditions where power and strength (Type II-dependent) are most impaired, the fiber-type effects of ActRIIB inhibition may be particularly beneficial.
Metabolic Effects Beyond Muscle Mass
While muscle hypertrophy is the most prominent effect of ACE-031, research has revealed broader metabolic consequences of ActRIIB pathway inhibition. Skeletal muscle is the body's largest metabolic organ and a primary site of glucose disposal and fatty acid oxidation. Increasing muscle mass therefore has systemic metabolic implications.
Studies in animal models have demonstrated that ActRIIB inhibition improves glucose homeostasis and insulin sensitivity, with reduced fasting glucose and improved glucose tolerance. These effects likely result from increased muscle mass providing greater capacity for glucose uptake and storage, though direct effects on muscle insulin signaling have also been proposed.
Fat mass reductions have also been observed with ActRIIB inhibition, potentially through increased metabolic rate associated with greater muscle mass. This body composition shift—increased lean mass with decreased fat mass—represents favorable metabolic remodeling with potential implications for obesity and metabolic syndrome research.
Safety Considerations in Research
The clinical development of ACE-031 provided important safety data that informs research applications. Key findings include:
Vascular Effects: The association with nosebleeds, gum bleeding, and telangiectasias reflects effects on BMP9/10 signaling, which is important for vascular endothelial function. These effects appear dose-dependent and were generally reversible upon treatment discontinuation.
Reproductive Considerations: Myostatin and related ligands play roles in reproductive tissues, and ActRIIB inhibition may affect reproductive function. Preclinical studies have explored these effects, though human data are limited.
Bone Effects: While increased bone mineral density was observed and could be beneficial, long-term effects on bone quality and remodeling require consideration. The interaction between muscle-derived signals and bone metabolism (the "muscle-bone unit") adds complexity to predicting outcomes.
Cardiac Considerations: As a muscle tissue, cardiac muscle could potentially respond to myostatin inhibition. While cardiac effects of ACE-031 appear minimal, this remains an important consideration for research protocols.
Research Applications and Methodology
For investigators studying ACE-031 and the myostatin pathway, several methodological considerations are important:
Biomarker Assessment: Serum myostatin levels can be measured to confirm target engagement, though interpretation is complicated by the fact that ACE-031 may increase total myostatin while decreasing free (active) myostatin. Measuring the ACE-031:myostatin complex may provide better assessment of target engagement.
Outcome Measures: Muscle mass can be assessed through various modalities including dual-energy X-ray absorptiometry (DEXA), magnetic resonance imaging (MRI), and computed tomography (CT). Functional assessments including strength testing, exercise tolerance, and daily activity measures complement anatomical outcomes.
Histological Analysis: Muscle biopsy allows detailed assessment of fiber size, fiber type composition, satellite cell numbers, and fibrosis—important parameters for understanding treatment effects at the tissue level.
Dosing Considerations: The long half-life of ACE-031 (approximately 10-14 days) allows for infrequent dosing, typically every two weeks in clinical studies. Dose-response relationships have been characterized, with muscle mass effects observed across a range of doses.
Future Directions and Scientific Impact
While ACE-031's clinical development was discontinued, the compound's scientific legacy continues to influence muscle biology research. Key ongoing areas of investigation include:
Next-Generation Inhibitors: The insights from ACE-031 development have guided creation of more selective myostatin pathway inhibitors designed to retain muscle-building effects while minimizing vascular side effects. These include both antibody-based and ligand trap approaches with modified binding profiles.
Combination Approaches: Research explores combining myostatin inhibition with other interventions including exercise, nutrition, and complementary therapeutics. Understanding how ACE-031 interacts with other muscle-building stimuli informs optimal therapeutic strategies.
Biomarker Development: The clinical experience with ACE-031 has contributed to development of biomarkers for assessing muscle mass and function, important tools for future clinical trials in muscle wasting conditions.
Pathway Elucidation: Research with ACE-031 continues to reveal nuances of TGF-β family signaling in muscle and other tissues, advancing fundamental understanding of this important signaling network.
Conclusion
ACE-031 represents a scientifically important research tool that has substantially advanced understanding of myostatin pathway biology and its therapeutic potential. As a soluble ActRIIB decoy receptor, ACE-031 demonstrated robust muscle-building effects that validated the concept of myostatin inhibition as a therapeutic strategy for muscle wasting conditions.
The clinical development journey of ACE-031, including both its successes and challenges, has informed the broader field of muscle therapeutics. The compound's muscle effects established proof-of-concept for ActRIIB inhibition, while the vascular findings highlighted the importance of ligand selectivity and guided development of next-generation inhibitors.
For researchers investigating muscle biology, regeneration, and therapeutic strategies for muscle wasting, ACE-031 remains a valuable tool for understanding the complex interplay between myostatin, satellite cells, and muscle homeostasis. Continued investigation of this pathway holds promise for addressing the significant unmet medical need represented by muscle wasting conditions affecting millions of patients worldwide.
References
- 1. Lee, S.J., et al. (2005). Regulation of muscle mass by myostatin. Annual Review of Cell and Developmental Biology, 20, 61-86.
- 2. Attie, K.M., et al. (2013). A single ascending-dose study of muscle regulator ACE-031 in healthy volunteers. Muscle & Nerve, 47(3), 416-423.
- 3. Campbell, C., et al. (2017). Myostatin inhibitor ACE-031 treatment of ambulatory boys with Duchenne muscular dystrophy: Results of a randomized, placebo-controlled clinical trial. Muscle & Nerve, 55(4), 458-464.
- 4. McPherron, A.C., Lawler, A.M., & Lee, S.J. (1997). Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature, 387(6628), 83-90.
- 5. Lee, S.J., & McPherron, A.C. (2001). Regulation of myostatin activity and muscle growth. Proceedings of the National Academy of Sciences, 98(16), 9306-9311.
- 6. Saitoh, M., et al. (2017). Myostatin inhibitors as pharmacological treatment for muscle wasting and muscular dystrophy. JCSM Clinical Reports, 2(1), 1-10.
All research information is for educational purposes only. The statements made within this website have not been evaluated by the US Food and Drug Administration. The statements and the products of this company are not intended to diagnose, treat, cure or prevent any disease.